Information Links
Related Conferences
Previous Issues Volume 4, Issue 1 - 2020
Ceftriaxone, an Empirical Goldmine: A Systematic Review of Randomized Controlled Trials
Mumtaz Shirin, Md. Shahidul Islam*
Department of Pharmacy, University of Science and Technology Chittagong (USTC), Chattogram, Bangladesh
*Corresponding author: Md. Shahidul Islam, Assistant Professor, Department of Pharmacy, University of Science & Technology Chittagong (USTC), Chattogram, Bangladesh, E-mail: [email protected].
Received Date: August 28, 2020
Published Date: October 01, 2020
Copyright: Islam MS. ©2020.
Citation: Islam MS, Shirin M. (2020). Ceftriaxone, an Empirical Goldmine: A Systematic Review of Randomized Controlled Trials. Mathews J Pharm Sci. 4(1):01.
ABSTRACT
Empiric therapy for community and health-care associated bacterial infections with high mortality is challenging with the continued development of resistant strains and the threat of multi-drug–resistant organisms. Pharmacists may impact patients’ outcomes by appropriately selecting initial antibiotic treatment, screening for duplication of therapy, and identifying the duration of therapy, regardless of whether a facility holds an antimicrobial stewardship program in place. The choice of ceftriaxone as an empiric therapy has been under-represented in many ways and its choice as first-line antibiotic in fatal infections remain controversial. To investigate the current state of ceftriaxone, alone or as a part of step-down therapy, therapy in terms of clinical and bacteriological efficacy, as well as evaluate the economic burden intravenous ceftriaxone therapy poses in patients with six infections associated with severe mortality and morbidity worldwide and assess the reliability of ceftriaxone as an empirical therapy in these six infections since the increased threat of multi-drug resistant organisms. This systemic review with meta-analysis of randomized controlled trials involves the assessment of the clinical and microbiological efficacy of ceftriaxone compared with that of other antibiotics in community-acquired pneumonia, bacterial meningitis, acute pyelonephritis, gonorrhoea, complicated intra-abdominal infections and efficacy in the perioperative prophylaxis of local and systemic infections published in the dates between1990 to 2019 was performed. The electronic databases of PubMed, the Cochrane Central Register of Controlled Trials and Google Scholar were reviewed to search for relevant randomized controlled trials. Additional references, review papers, and proceedings of seminars were also searched. The simplified once-daily administration of ceftriaxone, due to its long elimination half-life, compared to otherwise complicated regimens of other antimicrobials has given a superior advantage in a hospital setting as well as a in an ambulatory setting as a parenteral agent. The well-endowed tolerability profile and favorable pharmacokinetic profile has indeed cemented ceftriaxone as a drug of choice among the other cephalosporins in the treatment of both serious community-acquired and hospital-acquired infections.
KEYWORDS: Ceftriaxone; Review; Diseases; Efficacy; Cephalosporins
INTRODUCTION
In most instances where patients acquire infections in hospital, or are admitted to hospital having developed an infection at home, antibiotic therapy must be started before there has been time to determine the precise bacterial aetiology [1]. Other important subjects relevant to resolution concerning therapy are: surviving antibiotic policies, site of the infection, toxicity and drug interactions [2]. Consequently of considerations, a lot of clinicians prefer as the routine first-line treatment either the aminoglycoside or the third-generation of cephalosporin. The clinical pharmacists along with help of the infectious disease team have the integral responsibility in a everyday practice for judicious use of the antibiotics contained by a short time and promoting additional patient safety. A suitable antimicrobial consumption is a keystone for containment of the antimicrobial resistance in addition to good clinical as well as economic results [3]. There are considerable advantages to the shorter route of in-hospital management. The indirect costs of the admission to the hospital are very important restrictions for the poor families [4]. The preservation of intravenous admittance over the 10-day stage is very difficult as well as is the burden on limited hospital resources accessible in numerous developing countries. The suitable use of the antibiotics depends upon selection of the agent competent of achieving the desired serum deliberation to target presumed microorganism at site of the infection along with a satisfactory safety profile. Inappropriate in addition to unnecessary antimicrobial practice leads to the increase in the healthcare costs in addition to pathogen resistance [5]. Ceftriaxone is an IV antibiotic whose use has skyrocketed due to the advantages it pertains. Previous reviews have been conducted on ceftriaxone, it’s therapeutic efficacy in comparison to other drugs along with its tolerability profile by the ADIS Drug Evaluation (Drugs) three times but has recently lost its relevance since the last review in 2002 due to the approval of newer drugs in the market and increase in antibiotic resistance [6]. It is therefore important to reassess the place of ceftriaxone in the therapeutic regimens of severe and fatal infections compared to the existing empirical treatment, including the 4th and 5th generation cephalosporins, on account of the efficacy, safety and pharmaco-economics [7].
AIM OF THE REVIEW
A suitable antimicrobial consumption is a keystone for containment of the antimicrobial resistance in addition to good clinical as well as economic results. There are substantial advantages to a shorter course of in-hospital treatment. The appropriate use of antibiotics depends on the selection of an agent capable of achieving a desired serum concentration to target the presumed organism at the site of infection with an acceptable safety profile. Inappropriate and unnecessary antimicrobial usage leads to an increase in healthcare costs and pathogen resistance.
• The main purpose of this review is to give the clinical conclusion makers along with rationale for use of the ceftriaxone, either unaccompanied or as division of the step-down treatment, in management of the complex enduring cases in the hospital setting from admission through discharge. For inpatient management, it is important that such intravenous antibiotics be optimized compared to other intravenous antibiotics in terms of clinical efficacy, bacteriological eradication as well as the financial burden that might be posed in such patients.
• Another objective of this review serves the outpatient management. The use of ceftriaxone on an outpatient basis as an empirical agent in uncomplicated gonorrhoea and acute pyelonephritis has been underestimated. A secondary ambition of this review is to explicate the role of ceftriaxone in the management of such infections.
• The clinical and microbiological efficacy of ceftriaxone has been challenged in the recent years due to the recent development of Gram-negative organisms producing extended-spectrum β-lactamases (ESBLs). The susceptibility of such organisms has reduced to ceftriaxone as a result. Therefore, an additional aim is to evaluate the safety and efficacy of the “potential” anti-microbial agents compared to ceftriaxone, alone or as a part of step-down therapy.
In the previous reviews, the clinical and bacteriological efficacies were measures, although gaps of knowledge have been created in the recent years since changes in anti-microbial therapy, development of anti-microbial resistance, and the increase in the availability of ceftriaxone in developing countries since the release of its patent protection in 2005. Therefore, to address these gaps in knowledge, the following research questions were addressed:
• Is ceftriaxone inferior to the newly developed cephalosporins in terms of clinical efficacy, bacteriological eradication as well as tolerability?
• Is the use of ceftriaxone still permissible as an empiric therapy in the infections under study?
• Does ceftriaxone pose a greater economic burden on patients than other non-inferior anti-microbial agents?
• Does ceftriaxone use as a part of step-down therapy provide non-inferiority to other broad-spectrum antibiotics of similar efficacy? If so, what is the economic impact?
METHODOLOGY
Systematic reviews as well as meta-analyses are the essential instruments for the summarizing evidence precisely and reliably. They assist clinicians remain up to date; give evidence for the policy creators to the judge risks, advantages in addition to harms of the healthcare performances furthermore interventions; congregate together in addition to summarize related investigation for the patients in addition to their professions; give a starting position for the clinical practice instruction developers; give summaries of the previous investigation for the funders wishing to hold up new research furthermore help the editors judge and merits of the publishing statements of the new studies.
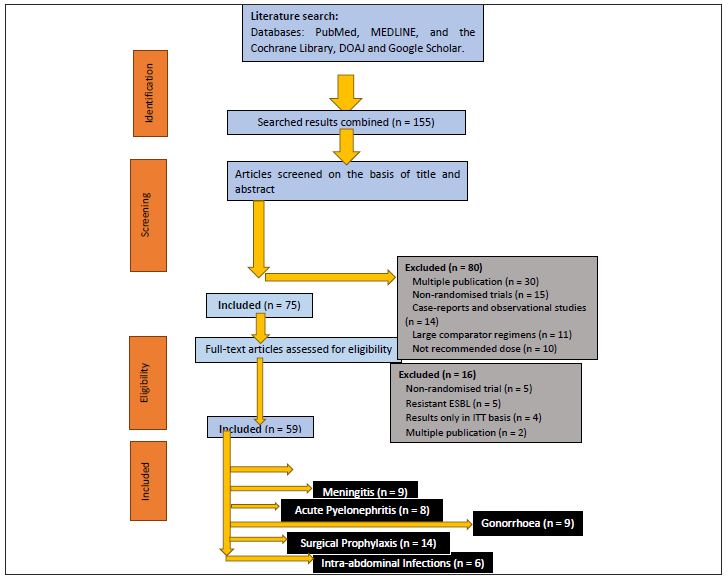
A methodical literature research was executed for the articles published previous to August 2020 following a pre-specified search policy in the PubMed, the nature, the Cochrane Central Register of Controlled Trials, the lancet, Directory of Open Access Journals, MEDLINE and Google Scholar to identify the relevant randomized controlled assessments. The United States Food and Drug Administration (FDA) along with European Medicines Agency profiles on the ceftriaxone, cefepime, and cefixime were used for comparison of bacteriological activity. The Clinical and Laboratory Standards Institutes 27th Edition of Performance Standards for Antimicrobial Susceptibility Testing M100 was used to compare the activity of ceftriaxone with cefuroxime, cefuroxime, cefixime, cefotaxime, cefepime and ceftaroline against some of the bacteria susceptible to ceftriaxone. We also hand searched the bibliographic references to conduct a profile on ceftriaxone and checked the reference lists on previously conducted reviews to correlate past and future data.
RESULTS
Ceftriaxone is the third generation new semisynthetic cephalosporin. It has a extensive half-life and has consequence in the advised once daily direction list, intravenously and intramuscularly in addition to has the broad spectrum of the activity against the Gram-positive as well as the Gram-negative bacteria, aerobic along with some anaerobic. Moreover activity of the ceftriaxone is commonly superior to that of first as well as second generation of cephalosporins in opposition to the Gram-negative bacteria excluding less than earlier generations of the cephalosporins in opposition to numerous Gram-positive bacteria. Ceftriaxone was very effective in obscured in addition to uncomplicated urinary tract infections and also the lower respiratory tract infections, bone, skin, soft tissue in addition to joint infections [8]. Except for diarrhea, ceftriaxone is safe, well tolerated, and not associatedwith the coagulopathies and disulfiram reactions reported with cefoperazone and moxalactam. The longhalf-life of ceftriaxone, which may allow for once- ortwice-daily administration, could have significant costand convenience benefits, making it the preferred agentof the class in some infections [9]. The communication of an significant antimicrobial drug, ceftriaxone along with zinc sulphate in addition to antacid calcium hydroxide were studied in aqueous system at the pH 7.4 by the variety of the physical method such as inspection of the spectral actions, the Job’s method of the continuous distinction by the spectrophotometry. At the pH 7.4 ceftriaxone appearances sturdy 1:1 complex along with zinc sulphate in addition to calcium hydroxide, indicated as opposite of “V” shaped curves that might indicate sturdy kinetics of the complexation. The trial samples in addition to the typical ceftriaxone disk investigated alongside Staphylococcus aureus. It was seen that antimicrobial activity of the ceftriaxone declines when it appearances complexes along with the ZnSO4.7H2O in addition to antacid solution such as calcium hydroxide. Moreover, by the antimicrobial examination it has been confirmed that zone of the inhibition of the ceftriaxone along with metals such as Zn and Ca condensed from 16 mm to the 14mm and 11mm reverentially [10]. In vitro antimicrobial activities of ceftriaxone and its complexes were tested using the reported methods. The chosen strains covered the Gram positive and Gram-negative bacteria as well as the antifungal activity. The data showed that the complexes has less biological activity than that of the ceftriaxone except the copper complex that gave the same activity towards most of the tested bacteria strains. In addition, Cu and Fe ceftriaxone complexes showed medium biological activity towards E. coli which is not affected by ceftriaxone drug [11]. Antibacterial drugs selected for clinical use vary widely by institution, region and country, the choice of agent being influenced by many factors, including the spectrum of antibacterial activity, local bacterial resistance patterns, pharmacokinetic characteristics, evidence of efficacy, tolerability profile, convenience of administration and cost. Before discussing all the uses of the ceftriaxone, this valuable review has spotlighted new applications of drug and bring up to date its relevance on the infections since last review in addition to its use in the infections and the contributing pathogens and their resistance models have varied over past decades. Since the last review on the application of ceftriaxone in bacterial infection, several bacteria have increased resistance as well as increased susceptibility against ceftriaxone [12]. Since the emergence of penicillin-resistant S. pneumoniae, the guidelines in the management of community acquired infections have gone through significant changes. Since the cephalosporins along with sturdy activity alongside S. pneumoniae, ceftriaxone and cefotaxime are important agents in these infections. Recent data on ceftriaxone deals with its activity in pneumococcal infections and the role it plays in case of penicillin resistance [13]. Ceftriaxone, unlike other antimicrobial agents, have shown fewer patterns on the development of microbial resistance to that parental use of the drug. The few cases that have been reported with these superbugs have been resolved by alternative cephalosporins that have a broader spectrum of activity such as cefepime and ceftaroline, and granting a scale-back on the use of ceftriaxone and other extended-spectrum cephalosporins that have caused the emergence of superbug that may lead to untreatable infections (gonorrhoea). Though compared to the other cephalosporins in the randomized control study, ceftriaxone, in the hospital setting, demonstrated emergence of the fewer cephalosporin-opposed to the Gram-negative bacilli in intestinal tract of the patients [14].
So third-invention cephalosporins offer action against the gram-negative life forms and reasonable the gram-positive action as well ability to penetrate into the CSF. The ceftriaxone generally gives equivalent and greater activity alongside these organisms contrasted with the presently marketed the third-generation of cephalosporins such as cefoperazone, ceftizoxime, cefotaxime, and moxalactam. Except for the diarrhea which in the most belongings has not required the therapeutic change and also ceftriaxone is very safe, healthy tolerated in addition to not connected with the coagulopathies and also disulfiram reactions accounted with the cefoperazone and also moxalactam [15]. On the whole, it was additional active against the P.mirabilis than the other compounds except some of the indole-positive Proteus [16].
CONCLUSION
Finally it can conclude that ceftriaxone has acquired its position in the therapy as first-line experimental agent, either unaccompanied or as ingredient of the combination treatment, in treatment of the invasive penicillin- opposed to the pneumococcal infections for the reason that it has the strong commotion alongside S. pneumoniae. The beginner's once-daily management of the ceftriaxone, owing to its long taking away half-life, contrasted to or else complicated treatments of other antimicrobials has provided as the superior improvement in the hospital setting in addition to the ambulatory setting such as parenteral agent. Moreover well-endowed permissibility profile in addition to complimentary pharmacokinetic outline has certainly cemented. So, ceftriaxone is as the drug of preference in the midst of other the third-generation cephalosporins in treatment of the both severe community-attained as well as hospital-attained infections.
REFERENCES
- Bradley JD, Wassel RT, Lee L, Niambar S. (2009). Intravenous Ceftriaxone and Calcium in the Neonate: Assessing the Risk for Cardiopulmonary Adverse Events. Pediatrics. 123(4):609-613.
- Herikstad H, Hayes PS, Hogan J, Floyd P, Snyder L, et al. (1997). Ceftriaxone-resistant Salmonella in the United States. Paedriatic Infectious Disease Journal. 16(9):904-905.
- Marhoum E, Noun M, Chakib A, Zahraoui M, Himmich H. (1993). Ceftriaxone versus penicillin G in the short-term treatment of meningococcal meningitis in adults. European Journal of Clinical Microbiology. 12:766-768.
- Rolfe RD, Finegold SM. (1982). Comparative In Vitro Activity of Ceftriaxone Against Anaerobic Bacteria. Antimicrobial Agents and Chemotherapy. 22(2):338-341.
- Laghari M, Memon AH, Mughal UR, Almani F. (2008). Spectrophotometric determination of ceftriaxone using 4-dimethylaminobenzaldehyde. Pakistan Journal of Analytical and Enviromental Chemistry. 9(1):1-7.
- Izadi M, Dadsetan B, Najafi Z, Jafari S, Mazaheri E, et al. (2018). Levofloxacin Versus Ceftriaxone and Azithromycin Combination in the Treatment of Community Acquired Pneumonia in Hospitalized Patients. Recent patents on anti-infective drug discovery. 13(3):228-239.
- Islam S, Rahman A, Al Mahmood AK. (2018). Bangladesh Pharmaceutical Industry: Perspective and the Prospects. Bangladesh Journal of Medical Science. 17(4):519-525.
- Hook EW, Golden MR, Taylor SN, Henry E, Tseng C, et al. (2019). Efficacy and Safety of Single-Dose Oral Delafloxacin Compared With Intramuscular Ceftriaxone for Uncomplicated Gonorrhea Treatment. Sex Trans Dis. 46(5):279-286.
- Ghose P, Islam SM. (2019). Ceftriaxone with Antacid and Metal Complexation and Investigation of Antimicrobial Activity, In-Vitro Demonstration. International Research Journal of Pharmacy and Medical Sciences. 2(4):1-7.
- Duncan CJA, Barr DA, Seaton RA. (2012). Outpatient parenteral antimicrobial therapy with ceftriaxone, a review. International Journal of Clinical Pharmacy. 34:410-417.
- Demirtas A, Yildirim YE, Sofikerim M, Kaya EG, Akinsal EC, et al. (2012). Comparison of Infection and Urosepsis Rates of Ciprofloxacin and Ceftriaxone Prophylaxis before Percutaneous Nephrolithotomy: A Prospective and Randomised Study. The Scientific World Journal. 2012:916381.
- Corrêa JC, Badaro R, Bumroongkit C, Mera JR, Dolmann AL, et al. (2003). Randomized, open-label, parallel-group, multicenter study of the efficacy and tolerability of IV gatifloxacin with the option for oral stepdown gatifloxacin versus IV ceftriaxone with the option for oral stepdown clarithromycin. Clin Ther. 25(5):1453-1463.
- Ceran C, Oztoprak I, Cankorkmaz L, Gumuş C, Yildiz T, et al. (2005). Ceftriaxone-associated biliary pseudolithiasis in paediatric surgical patients. Int J Antimicrob Agents. 25(3):256-259.
- Cannavino CR, Nemeth A, Korczowski B, Bradley JS, O'Neal T, et al. (2016). A Randomized, Prospective Study of Pediatric Patients With Community-acquired Pneumonia Treated With Ceftaroline Versus Ceftriaxone. Pediatr Infect Dis J. 35(7):752-759.
- Cummins LH, Kozak PP, Gillman SA. (1976). Theophylline determinations. Ann Allergy. 37:450–451.
- Rolfe RD, Finegold SM. (1982). Comparative In Vitro Activity of Ceftriaxone Against Anaerobic Bacteria. Antimicrobial Agents and Chemotherapy. 22(2):338-341.