Previous Issues Volume 2, Issue 1 - 2017
Calorimetric Evaluation of Amoxicillin Stability in Aqueous Solutions
Ervin Palma1 , Lonny Ellison2 , Elias Meza2 , Yuri Griko3
1Evergreen Valley College, San Jose, CA 95135, USA.
2 Carnegie Mellon University Silicon Valley, Moffett Field CA 94035, USA.
3 National Aeronautics and Space Administration, Ames Research Center, Moffett Field, CA 94035 USA.
Corresponding Author: Yuri Griko, Space Biosciences Division, NASA Ames Research Center MS 261-3, Moffett Field, CA 94035, USA.
Tel: 650-604-0519; E-Mail: [email protected]
Received Date: 03 Jun 2016 Accepted Date: 21 Jun 2016 Published Date: 11 Aug 2016
Copyright © 2016 Griko Y
Citation: Palma E, Ellison L, Meza E and Griko Y. (2016). Calorimetric Evaluation of Amoxicillin Stability in Aqueous Solutions. Mathews J Pharm Sci. 2(1): 008.
ABSTRACT:
The thermal properties of amoxicillin suspension in aqueous solutions were analyzed by differential scanning calorimetry under various solvent conditions to identify changes in thermodynamic parameters closely related to drug absorption and pharmacokinetic behavior.
Analysis of thermal profiles of amoxicillin in solid form and in aqueous suspension showed several phase transitions, not reported previously, which correspond to different transformations in drug integrity induced by temperature. The low temperature endothermic transition in temperature range 20-50°C is described as associated with decomposition of crystal-like structure of amoxicillin in liquid suspension, which cooperatively dissembles with increase of temperature. The low temperature endothermic transition is following exothermic transition in temperature range 60-125°C with a pH dependent temperature maximum occurring in 80-114°C range. While the maximum temperature for the endothermic transition is practically independent of the solution pH, the exothermic transition displays strong pH dependence, decreasing its Tmax as the pH of solution decreased from pH 7 to pH 2.0. During heating of the amoxicillin suspension the exothermic transition demonstrates a complex character with several maximums occurring on the calorimetric thermogram. The physical and chemical changes in the amoxicillin are associated with the degradation of amoxicillin, as evident by the yellowing of the solution as it is heated.
The temperature induced changes observed for the amoxicillin aqueous suspension contrast those reported for the solid form of amoxicillin where only endothermic thermal transition in temperature range 55-125°C are observed. The latter is associated with dehydration loss crystallinity of the solid form. Based on comprehensive calorimetric analysis, this manuscript describes the thermal properties as well as transformations between the different states of amoxicillin during heating.
KEYWORDS:
Amoxicillin; Stability; Thermal Transitions.
ABBREVIATIONS:
DSC - Differential Scanning Calorimeter; UV - Ultra Violet; Na-P - Sodium Phosphate; Na-Ac - Sodium Acetate; Gly-HCl - Glycine Hydrochloride; Tmax - Temperature of Peak Maximum; ΔH - Enthalpy Change; pH - pH of a solution, the molar concentration of hydrogen ions; I - Ionic Strength of a Solution; M&M - Materials and Methods.
INTRODUCTION:
Amoxicillin trihydrate is a beta lactam antibiotic that has been used worldwide to treat a broad spectrum of bacterial infections including the upper respiratory tract, cystitis, peritonitis, gonorrhea, intra-abdominal sepsis, skin and soft tissue infections [1]. The antibiotic formulations are available in various solid and liquid dosages consisting of numbers of excipients to improve its effectiveness. Reconstitution of amoxicillin powder for the injection of high doses (250mg, 500mg and 1g) is considered when the oral route is unsuitable or urgent treatment of severe infection is required [2]. Unfortunately, due to the wide variability in the quality of drugs in solid form, their dissolution and pharmacokinetic profiles may behave unpredictably under conditions which differ from those tested by the manufacture [3]. Most of the drugs developed in the pharmaceutical industry have poor solubility or are practically insoluble in water. The low aqueous solubility may result in new chemical entities which are usually associated with poor parental and oral bioavailability [4]. Upon reconstitution in aqueous solution some fraction of drug is solubilized while another one remains in highly hydrated insoluble forms at concentrations above the drug's solubility point. Although, the description of the thermal behavior of amoxicillin solid polymorphs has already been given in the literature, their thermodynamic characteristics in aqueous suspension are not well clarified. For the category of poor soluble drugs that require reconstitution from solid form to the injectable liquid, determination of physicochemical properties, thermodynamic stability and inter-conversion conditions are essential for optimization of drug absorption and pharmacokinetics. It has been noted that differences in solubility may affect antibacterial activity more than pharmacokinetics [5]. Therefore it is important for pharmaceutical practice to know stability and physical properties of drug molecules and their interactions forming the heterogeneous mixture of drug suspension to predict its pharmacokinetic and pharmacodynamic behavior. The presence of insoluble forms may impact drug permeability at the site of absorption. The nature of the physical forms of amoxicillin, which belongs to the category of poorly soluble drugs, plays a key role in affecting its dissolution from a solid dispersion when given orally in high doses or parentally.
The aim of this study, therefore, was to investigate specific changes in the structural arrangement of amoxicillin in aqueous suspension at different temperatures and to characterize the physical and chemical states of the drug.
MATERIAL AND METHODS:
Preparation of Tablets and Physical Tests
Amoxicillin trihydrate (Teva Pharmaceuticals, USA) was provided as a 500mg encapsulated dry powder by the Medical Unit of NASA Ames Research Center (Moffett Field, CA). The molecular mass of amoxicillin trihydrate MW = 365.4 g/mol has been used in this analysis. All reagents used in this study were of analytical grade (Sigma-Aldrich, Germany). Gly-HCl buffer was used for preparation of samples with pH 2 and pH 3. Buffers with pH 4 and pH 5 were prepared from sodium acetate. Buffers at pH=7 and pH=8 were prepared from sodium phosphate. The pH of buffers and solutions of amoxicillin trihydrate were measured using a pH meter (IQ Scientific Instruments, San Diego, USA), calibrated with buffer standards with error ± 0.02 pH units.
Amoxicillin powder was reconstituted in select buffer solutions (pH 2-8) at ambient temperature and shaken for an hour to homogenize the amoxicillin preparations [6]. The preparation was considered as homogeneous following visual evaluation. Reconstituted samples can be stored at 4°C temperature over period of 7 days without any detectable degradation or changes in solubility as assayed by UV-spectroscopy [7]. For solubility testing of amoxicillin at different pH values, the suspensions were filtered through Milipore filter with pore size 0.45mm and its amount of soluble fraction was determined spectrophotometrically using the molar extinction coefficient of amoxicillin at 254 nm (?AM = 1050 M-1 cm-1) [8]. Lambda 35 UV- double beam recording spectrophotometer (PerkinElmer Inc.) with a 1 mm length quartz cuvette were used for all absorbance measurements. The concentration of amoxicillin was corrected for light scattering when measured spectrophotometrically. Its value before and after filtration enables a precise measurement of the soluble and insoluble fraction under each given solvent conditions.
Calorimetric experiments were performed using a Microcal VP-DSC microcalorimeter with a heating rate of 1 deg C°/min and external pressure 32 atm.
The amoxicillin concentration in calorimetric experiments varied in the range of 0.625mg/ml - 14 mg/ml depending on conditions of each experiment.
The amoxicillin in solid powder form was analyzed using conventional Q100 DSC (TA Instrument, USA). Approximately 2 to 5 mg of each sample was heated in an open aluminum pan from 30 to 250°C at a scanning rate of 10°C/min under a stream of nitrogen gas.
An optical microscope Axioskop 2 (Carl Zeiss Microscopy, LLC, USA) with a digital camera attached to a PC utilizing software was used to record the images of amoxicillin samples spreading onto glass slabs
RESULTS:
Figure 1 shows the calorimetric thermogram for amoxicillin reconstituted in 20 mM sodium phosphate buffer, pH 7.0. The DSC scan of liquid amoxicillin suspension at concentration 5 mg/ml exhibits two major heat capacity peaks after heating to 130°C with the endothermic heat capacity peak in temperature range 25-40°C and exothermic peak centered at 110°C.
Both, endothermic and exothermic transitions are irreversible when heated to the temperatures of their completions (to 50°C for the endothermic transition, and to 130°C for the exothermic transition). In the temperature range of the endothermic transition none of the yellowish color development associated with amoxicillin degradation was observed. However, yellowing of the solution becomes evident after completion of the exothermic transition indicating that amoxicillin undergoes some chemical degradation in the temperature range of 50°C to 130°C.
Figure 1: Original DSC scans of amoxicillin in solution of Na-P buffer, pH 7.0 (a); scan after first heating of amoxicillin sample to 130°C indicating irreversibility (b), and reference scan in the same Na-P buffer with 2 mCal/ min calibration power pulse (c). Concentration of amoxicillin is 5 mg/ml.
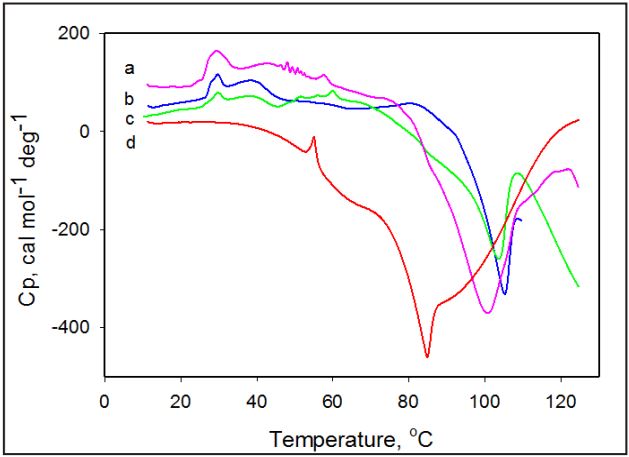
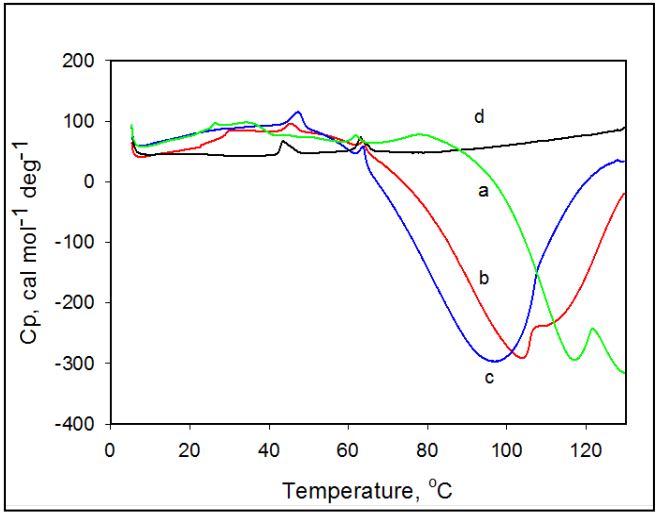
Effect of pH Thermally induced transformations in amoxicillin observed in temperature range 25°C-130°C are found to vary as a function of pH. Figure 2 shows thermograms of amoxicillin recorded in solutions at different pH. The exothermic heat capacity peak maximum shifts to lower temperatures and the heat capacity profile becomes broader as the pH of solution decreases. The broadness and appearance of shoulders in the heat capacity profile at low pH values indicates that the exothermic transition is complex and made up of several thermal transitions.
Figure 2: Temperature dependence of the partial heat capacity of amoxicillin in solution at different pH values. 20 mM Na-Ac, pH 5.0 (a); 20 mM Na-Ac, pH 6.0 (b); 20 mM Na-P, pH 7.0 (c); 20 mM Gly-HCI, pH 2.0 (d). Concentration of amoxicillin is 5 mg/ml.
The exothermic heat capacity peak maximum shifts from 105.2°C to 84.7°C as the pH of the solution decreases from 7.0 to 2.0. This can be explained by the fact that ionized forms of amoxicillin in acidic solution have weaker intermolecular interactions, which results in quicker decomposition for the samples with the lower pH [9]. Therefore, kinetics of degradation of amoxicillin is much faster in acidic solutions below pH 5 [10].
In contrast to the exothermic transition in amoxicillin, the temperature of the endothermic peak maximum remains unchanged by variation of pH of the solution. Although decreasing the pH of solution does not affect the temperature of the endothermic peak maximum it does lead to a decrease in the enthalpy of this transition, as made evident by the decrease of the area of the peak. The changes in pH of amoxicillin suspension from pH 5.5 result in increasing population of ionized drug forms and therefore solubility. The enthalpy of the endothermic peak associated with the loss of insoluble amoxicillin decreases. At pH values below pH 2.0 the endothermic transition becomes practically undetectable, while the exothermic transition remains present. A Similar effect of pH on amoxicillin thermal stability was also reported for its crystalline powder and interpreted as resulting from a decrease in intermolecular forces stabilizing the crystalline structure and therefore leading to a quicker decomposition for the samples with the lower powder pH [9, 11]. Although initially observed for powder, it is also applicable to highly hydrated crystals in aqueous solutions.
EFFECT OF IONIC STRENGTH:
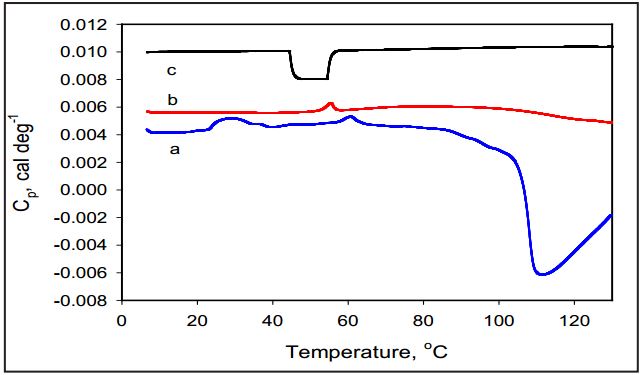
The overall stability of amoxicillin and its temperature induced molecular changes are also greatly influenced by variation in ionic strength of solution due to the underlying chemical structure of amoxicillin. Figure 3 shows that the set point temperature of the exothermal peak decreases from 125°C to 95°C, as the ionic strength of Na-P buffer increased from 20 mM to 400 mM. The enthalpy of the exothermic transition of amoxicillin at low ionic strength is difficult to estimate since the exothermic transition is not completed at 130°C. However, the shift of the Tmax of the exothermic transition with increasing ionic strength allows this process to be completed and to calculate the enthalpy of the transition. Results depict that enthalpy of the exothermic transition is -77.67 J/g in 200 mM Na-P increasing to ΔH= -109.32 J/g in 400mM Na-phosphate.
Figure 3: Temperature dependence of partial heat capacity of amoxicillin in solutions of Na-P buffer with different molarity. The molarity of the buffer are: 20 mM (a); 200 mM (b); 400 mM (c); scan after first heating indicating irreversibility of the process (d). Concentration of amoxicillin is 5 mg/ml.
In contrast to the exothermic transition, the Tmax of the endothermic transitions centered in temperature range 30-50°C does not depend significantly on the ionic strength of the buffering solution. At the same time the enthalpy of this transition does decrease substantially with increase in ionic strength and become undetectable in the 400 mM solution.
Taken together, these results demonstrate that variation in electrostatic interactions, underlying changes (effects) of pH and ionic strength of solution, differently affect the thermodynamic and structural stability of amoxicillin as evident by characteristics of temperature induced transitions in the considered temperature regions. The latter is associated with decrease in amount of insoluble material with increase of ionic strength of solution, made evident by visual observation.
EFFECT OF CONCENTRATION:
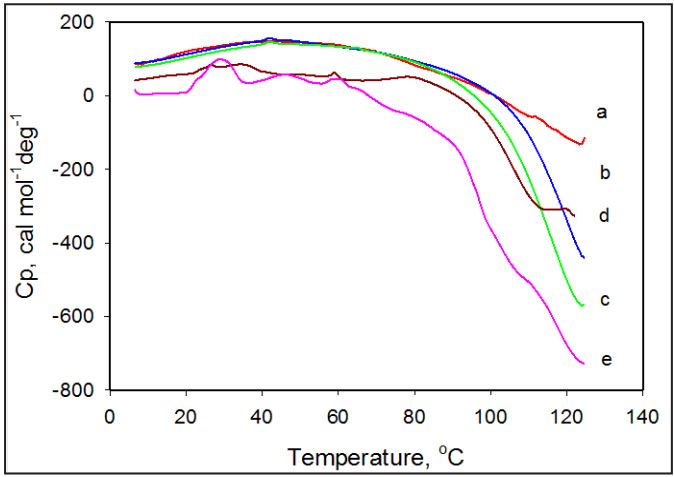
To further characterize the origin of the endothermal and exothermal transitions in the amoxicillin suspension, we performed calorimetric experiments with different drug concentrations below and above its solubility level. Increasing the amoxicillin concentration resulted in an increase in the enthalpy and changes in the shape of the endothermic peak (Figure 4). The concentration dependent complexity of the thermal profile may be caused by the presence of different forms of insoluble material which undergoes transformation at different temperatures within the temperature range of endothermal transition. Variation in amoxicillin concentration results in a re-distribution within the population and fraction of insoluble forms; which are different in size, intermolecular arrangements, and interaction with solvent. As a result the complexity of the endothermal heat capacity profile is a result of their (insoluble forms) fractional contribution in the heat capacity function. The observation that increase in the enthalpy does not substantially affect the set point temperature of the endothermic transition further indicates that the structure of insoluble material have different but close thermal stability regardless of the aggregates size. The increase enthalpy in the endothermic transition with amoxicillin concentration as a result of increase in population of the insoluble fraction, also affects enthalpy of the exothermic transition which occurs in temperature range 70-130°C. It is reasonable to suggest that after the decomposition of their intermolecular structure at the first endothermic transition, amoxicillin molecules from the melted crystals will join the association of soluble fraction to undergo further chemical degradation in temperature range 70-130°C.
As the concentration of amoxicillin is decreased below its maximum solubility (~4mg/ml), the intensity of the endothermic peak decreases and becomes undetectable at lower concentrations; however the exothermic transition still remains apparent on the DSC thermogram (Figure 4).
Figure 4: DSC thermogram of amoxicillin recorded at different concentrations of the drug. Values of concentrations in mg/ml are: 0.625 mg/ml (a); 1.125 mg/ml (b); 2.5 mg/ml (c); 5mg/ml (d); 7.5 mg/ml (e).
In order to determine the contribution of the insoluble fraction of the amoxicillin suspension to the calorimetric profile of melting, the suspension was filtered through the 0.45mkm filter to remove insoluble material. The low temperature endothermal transition, which is evident in all preparations of amoxicillin in the pH range 7-3 was no longer observable in filtrated samples (not shown). Removing the insoluble particles by filtration does not affect the enthalpy of the exothermic transition and provides clear evidence that the endothermic transition is associated with a fraction of insoluble forms in amoxicillin suspension.
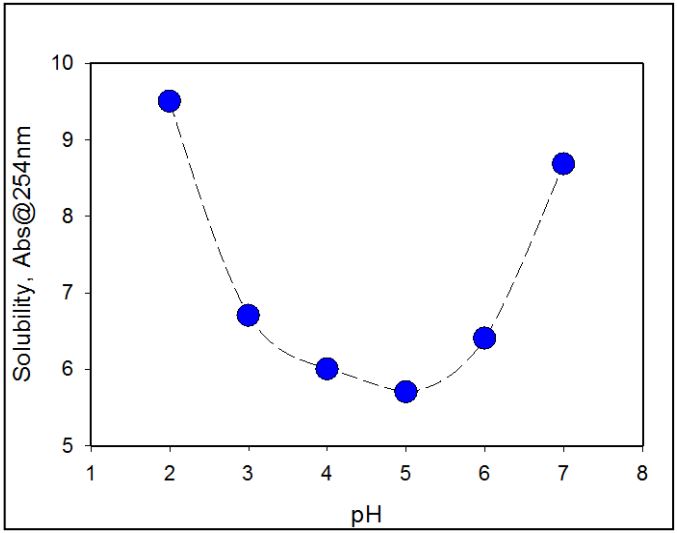
Figure 5 shows dissolution profile of amoxicillin trihydrate in the pH range 7-2 because the solubility of amoxicillin is a function of pH and of the ionic strength, changing the pH or/and ionic strength of solution toward less soluble conditions leads to changes in fraction of insoluble amoxicillin and enthalpy of the endothermic transition (Figure 2 and Figure 3).
Figure 5: Solubility of amoxicillin in solutions with different pH values as measured by measuring its absorption at 254 nm after filtering out the insoluble fraction (see M&M for details).
As a result, thermodynamic analysis of amoxicillin suspension at different concentrations and solvent conditions (pH, I) allows one to estimate the relative contribution of soluble and insoluble fractions present under each given solvent condition.
In order to determine the morphology of the insoluble forms of the amoxicillin suspension which are associated with the endothermic transition, samples were imaged under different conditions using optical microscopy. Figure 6 shows images of untreated amoxicillin trihydrate powder and amoxicillin suspension in 20mM Na-P buffer, pH 7.0 treated by filtration and by heating. It is evident that there are many crystals in the saturated aqueous suspension of amoxicillin (5mg/ml) that closely resemble those found in crystals in amoxicillin powder and not disordered amorphous aggregates. (Figure 6 a, b). There are no visible crystals after filtration of the amoxicillin suspension through the 0.45 µm filter as well as after heating to 60°C (Figure 6 c, d). These images demonstrates that the insoluble forms of amoxicillin in aqueous suspension appear to have a crystalline structure which undergoes transformation to soluble forms in cooperative-like manner when the temperature is increased.
Figure 6: Optical images of amoxicillin trihydrate crystals in solid state and aqueous suspension using optical microscope. (a) Amoxicillin untreated solid powder; (b) Amoxicillin suspension (5 mg/ml in 20 mM Na-P buffer, pH 7.0); (c) Amoxicillin suspension filtered through the 0.45 mkn Amecon filter; (d) Amoxicillin suspension heated to 60°C.
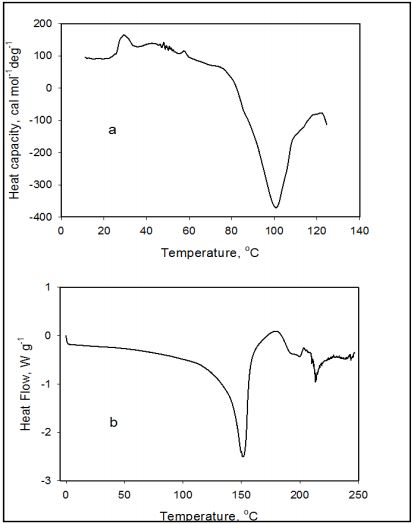
Comparison of Liquid and Solid Forms Figure 7 shows comparison of DSC thermograms for liquid and solid formulations of amoxicillin at pH 5.0. The Calorimetric profiles of amoxicillin in solution are different from those in the solid state. The endothermic transition of amoxicillin in solution occurs between 30°C and 55°C and is followed by a broad exothermal transition with Tmax centered at 100°C. The exothermic transformation appears to have a complex character and is further associated with chemical degradation of the amoxicillin molecule as followed from the yellowing of the solution. In contrast, the heat capacity function of amoxicillin in the solid state shows no endothermic transition in the temperature range between 30-55°C however exhibiting an endothermic transition (ΔH = 240.5 J/g) at a temperature 139.46°C. This was followed by the crystallization of amoxicillin trihydrate molecules at 180.4°C and immediately followed by the endothermic transition of a melting phenomenon started at 194°C which has been followed by the degradation of the amoxicillin molecules [11]. The endothermic transition of the solid forms of amoxicillin has been described in previous studies as to be related to the breaking of an intramolecular Hbonding network including a dehydration of crystal hydrates in this temperature range [11]. The enthalpy of the exothermic peak was estimated ΔH = -43.58 J/g at 180.4°C what is close to ΔH = -47.06 J/g found in previous studies [11, 12].
Figure 7: DSC thermal profile of amoxicillin in aqueous preparation as recorded with DSC-VP (a) and in solid state as recorded with DSC Q100 (b). pH of aqueous and solid preparation is 5.0. Mass of solid amoxicillin in calorimetric cell is 3.2 mg. Mass of amoxicillin in aqueous preparation in calorimetric cell is 2.5 mg.
DISCUSSION:
Stability of Crystal-like Forms in Amoxicillin Suspension: The results presented in this study demonstrate that at concentration above solubility level (> 4 mg/ml) or pH and ionic strength of solution when insoluble fractions is highly populated, DSC thermogram shows two heat capacity peaks associated with changes in amoxicillin induced by temperature. One of endothermic peaks in temperature range 30°-50°C strongly depends on the population of the insoluble fraction of amoxicillin molecules and is associated with the presence of stable, crystal-like particles of amoxicillin in aqueous solution, which "melt" cooperatively with an increase in temperature. The other peak centered between 80°-120°C depending on pH of solution is exothermic in nature and is associated with the chemical degradation of the amoxicillin molecules. As a result, the complexity of the heat capacity profile of amoxicillin reconstituted from solid powder depends on the populations of the soluble and insoluble fractions. It is remarkable that this transition still remains detectable at concentration - 1.8 mg/ ml which is below the solubility level (4 mg/ml), though its enthalpy is small. Filtration of the tested solution via 0.45 µm Millipore filter makes this peak undetectable on the thermogram that exhibits only one exothermic peak centered at 80°- 120°C.
It is remarkable that the insoluble fraction of amoxicillin in solution still exhibits features of the highly ordered crystals found in the unsoluated powder (Figure 6). Small differences in morphology, size, and shape of the water stable crystals in suspension from the solid crystals could be due to the influence of water as a most important factor of their stability and integrity under a variety of solvent conditions. The ordered structure of the amoxicillin particles in water suspension is also evidenced by the sharpness (cooperativity) of the endothermic transition associated with disruption of their structure induced by temperature (Figure 7).
Both the crystal state of amoxicillin powder and insoluble particles in liquid suspension is that both of these structures are stabilized by positive enthalpy i.e. melts with endothermic heat effect. In the case of the crystalline powder, the endothermic peak on the DSC thermogram has been commonly interpreted to be associated with both dehydration and a loss of hydrate crystal from amoxicillin trihydrate [11, 13]. Furthermore, dehydration contributes positively to the endothermic effect of crystal loss, this can help to explain a higher enthalpy value in the case of amoxicillin powder than enthalpy of crystal description in the water environment. The enthalpy of intermolecular interactions stabilizing crystal structure is expected to decrease significantly in the aqueous suspension due to the effect of extensive water hydration and destabilization of the crystal form. Indeed, by making a correlation between highly hydrated "liquid" and dry crystals of amoxicillin, the enthalpy of the endothermic transition of amoxicillin in suspension is significantly smaller than the endothermic enthalpy associated with disruption of crystal structure in solid state. Since the population of the crystal-like fraction in solution depends on pH, (Figure 2), ionic strength (Figure 3) and concentration of amoxicillin (Figure 4), the enthalpy of this process depends on all those variables as well. The relative population of soluble and insoluble fraction of amoxicillin in suspension under different solvent conditions can be estimated from the amoxicillin solubility curve (Figure 5) to normalize enthalpy value. The enthalpy of amoxicillin associated with disruption of insoluble crystal-like particles was found to be ΔH = 17 J/g which is smaller than ΔH = 118.6-205 J/g estimated for loss of the crystal structure in solid amoxicillin trihydrate powder [11, 14]. The possible explanation for the high enthalpy in solid form of amoxicillin can be the positive enthalpy of dehydration accompanying crystal disruption, which contribute to stability of solid crystal forms of amoxicillin but absents in the case of liquid forms. As a result, the stability of the insoluble crystalline forms in a water environment is expected to be lower than for amoxicillin in solid crystal state due to the activity of bulk water molecule leading to a decrease in intermolecular interactions between drug molecules [15]. The comparison of Tmax for the endothermic transitions of amoxicillin in solid and liquid formulations (Figure 7) clearly demonstrate the difference in their stability. Crystalline structures in aqueous suspension melt at lower temperature (30°-50°C range) than crystalline structure in solid powder (80°-160°C range).
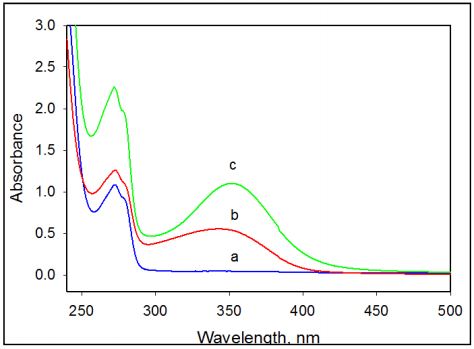
Exothermic Transition Associated with Amoxicillin Degradation in Aqueous Solution: According to the DSC thermogram, amoxicillin at concentrations below 4mg/ml displays only one exothermic peak centered at 80-120°C. This exothermic transition is associated with chemical degradation of the amoxicillin molecule induced by temperature. Spectrophotometric measurements of the absorption UV- spectra of amoxicillin show that in this temperature range amoxicillin undergoes change similar to what was observed for the degraded amoxicillin after 14 days of storage at 25°C. (Figure 8). The absorption in the UV spectrum at 254 nm progressively increases when amoxicillin undergoes chemical degradation accompanied by a developing yellowish color. Similar changes in the amoxicillin spectrum occur after heating the amoxicillin solution from 80°C to 120°C, the temperature range of the exothermic transition.
Figure 8: UV-spectrum of intact and degraded amoxicillin in 20 mM Na-P buffer, pH 7.0. (a) fresh aqueous preparation of amoxicillin; (b) amoxicillin degraded by heating to 130°C; (c) amoxicillin degraded by prolonged storage at 25°C for 14 day. Concentration of amoxicillin is 5 mg/ml.
The calorimetric measurements presented provide clear evidence that the degradation of the amoxicillin molecule in aqueous solution is accompanied by a negative enthalpic effect i.e. the reaction is exothermic. The temperature at which this process occurs and the value of enthalpy depends strongly on the solvent pH and ionic strength. This is opposite to what has been reported for the solid form of amoxicillin where the chemical degradation of amoxicillin described as an endothermic process [16]. Our observation that the exothermic transition on the calorimetric curve of liquid amoxicillin sample is associated with the chemical degradation of amoxicillin is supported by previous indirect measurements of the degradation kinetics of amoxicillin in solution which has also claimed the exothermic nature of the degradation process [17].
These results provide a clear evidence for a distinct energetic differences associated with structural change in amoxicillin occurring in solid state and in solution induced by temperature. The observed difference in the thermal behaviour of amoxicillin in solid and liquid states is associated with intermolecular interactions between molecules of amoxicillin which are radically different in its crystalline solid state relative to that obtained in a crystal state in solution. This is supported by the fluorescence study of amoxicillin which revealed a significant decrease in the energy of the excitation and emission bands of the solid state, relative to those of the free molecules in solution [18]. Similar reduction in energy transfer of the excitation and emission bands is observed during the heating of amoxicillin solutions to 55°C. This might be related to the decrease in cooperative inter-molecular interactions in the crystalline state during heating to high temperature resulting from the increasing entropic factor. The increase in entropy of internal intermolecular interactions stabilizes the crystallike structures of amoxicillin in solution with the elevation of temperature, resulting in a disruption of these cooperative interactions and in the dissociation of the molecular complexes. The latter also leads to an increase in solubility of amoxicillin at high temperature.
Link to Pharmacokinetics of Amoxicillin Suspension:It is unknown at this point whether the observed irreversible alterations in the thermodynamic characteristics of amoxicillin in the temperature range marginally to the physiological body temperature, may correlate with reported instability of particular amoxicillin formulation at physiological temperature.
After reconstitution of the solid powder in aqueous solution amoxicillin in a dosage form at a concentration above its solubility limit will exist as a suspension in two states, soluble and insoluble. This two-phase system will exhibit more complex characteristics and behavior than pure soluble and solid forms. Solid particles in suspension may retain more or less their original crystal structure similar to the hydrated solid states of drugs, and therefore may also have different polymorphs. The latter may significantly affect the absorption and degradation kinetics of drugs and have the same multi challenges that polymorphs may cause [19]. A dose dependency for amoxicillin absorption was observed with a lower than expected mean maximum plasma concentration. Zero-order kinetics of absorption was apparent along with a first order kinetics [20]. Non-linearity of the amoxicillin absorption kinetics indicates involvement of a number of factors in addition to simple diffusion and is of a practical significance. The presence of the stable insoluble amoxicillin particles in solution at intestinal pH and physiological temperature is the reason for the dose-dependent bioavailability of this drug, delaying the time to peak plasma concentration and decreasing the fraction absorbed. Intestinal absorption kinetics of amoxicillin is a zeroorder process, common for drug suspension, when solubility is the limiting factor [21]. At high concentrations, the degradation kinetics of amoxicillin in aqueous solutions also shows a deviation from the first order degradation kinetics, which is indicative of rate of multimerization reaction [22].
Characterization of the structural and thermodynamic properties of the stable forms of medications in drug suspensions may provide valuable information for precise monitoring of state and complexity of liquid formulations.
ACKNOWLEDGMENTS:
The authors acknowledge and appreciate the technical support by team members of the Medical Unit of NASA Ames Research Center (Moffett Field, CA) for providing commercial samples of amoxicillin.
REFERENCES:
-
www.mhra.gov.uk/home/groups/documents/websiteresources/con046543.pdf
-
http://cpharm.vetmed.vt.edu/VM8784/antimicrobials/ principles/principles.cfm
.jpg)