Previous Issues Volume 1, Issue 1 - 2016
Anti-Melanoma Agents Derived from Fungal Species
Anna Kilimnik1 , Valery M Dembitsky2
1National Institute of Water and Atmospheric Research, Private Bag 14-901,Wellington6241, New Zealand.
2Institute for Drug Discovery, P.O. Box 45289, Jerusalem 91451, Israel.
Corresponding Author: Valery M Dembitsky, Institute for Drug Discovery, 8 Ha-Marpe Str., P.O. Box 45289, Jerusalem 91451, Israel,
Tel: +972-526877444; E-Mail: [email protected]
Received Date: 27 Feb 2016 Accepted Date: 03 Mar 2016 Published Date: 16 Mar 2016
Copyright © 2016 Dembitsky VM
Citation: Dembitsky VM and Kilimnik A. (2016). Anti-Melanoma Agents Derived from Fungal Species. Mathews J Pharm Sci. 1(1): 002
ABSTRACT:
Ozone depletion by anthropogenic gases has increased the atmospheric transmission of solar ultraviolet-B radiation. There is a logical link between the natural defenses of terrestrial and marine organisms against UV radiation and the prevention of UV-induced damage of human skin. UV light degrades organic molecules such as proteins and nucleic acids, resulting in structural changes that directly affect their biological function. Compounds that block UV can be potentially developed for human use. The biological roles of more than 130 natural sunscreens, tyrosine and melanin inhibitors as a defense against solar radiation in fungal species, together with their structure, distribution, regulation, and effectiveness are reviewed.
KEYWORDS:
Anti-melanoma; Tyrosinase; Inhibitors; Sunscreens; Fungi; Fungal Endophytes; Lichens.
INTRODUCTION:
Short wavelength ultraviolet (UV) solar radiation has been shown to reach ecologically significant levels in terrestrial, freshwater, and marine ecosystems. Drastic stratospheric ozone depletion over the Antarctic and Arctic, as well as a moderate decrease in the total ozone column over high and mid-latitude water, has recently been reported [1-6]. Some recent reviews on various aspects of UV effects on terrestrial and aquatic ecosystems have been published [7-12]. The exposure to UV radiation originating from sunlight can cause harmful effects on human health, such as erythema (sunburn), melanoma (skin cancer), and premature skin aging. These effects are caused by UVA (320 nm to 400 nm) and UVB (290 nm to 320 nm) radiation. Despite the availability of diverse choices of sunscreens, the search for new and improved sunscreens and tyrosine and melanin inhibitors continues [13-16]. Melanoma is the most aggressive form of skin cancer and a serious health problem worldwide because of its increasing incidence and the lack of satisfactory chemotherapy for late stages of the disease [17]. Anti-melanoma agents (AMA) have been isolated from many natural sources, including plants, cyanobacteria and algae, and marine organisms [18-25].
Tyrosinase is a copper-containing enzyme, also known as polyphenol oxidase, which is widely distributed in microorganisms, plants, and animals. In human beings, tyrosinase is a key enzyme for melanin biosynthesis and is associated with melanin hyper-pigmentation and melanoma [26,27].
This review describes natural melanin production inhibitors (MPI), AMA, and UV-protecting sunscreens that have been discovered from fungi, lichenized fungi, and fungal endophytes.
Kojic acid and its analogues and derivatives:
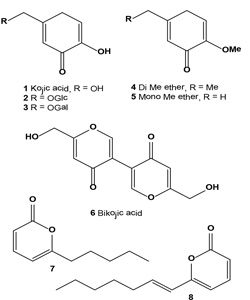
Kojic acid (1) was discovered in 1922 as a natural product produced by several species of fungi, especially Aspergillus oryzae, which has the Japanese common name koji and it is also produced by many species of Aspergillus, Acetobacter, and Penicillium [28,29]. It has been successfully used to lighten pigment spots and skin discoloration and is considered a popularly specialized inhibitor for melanin. At present, it is used in various types of cosmetics for curing freckles, spots on the skin of elder men, pigmentation, and acne. Kojic acid is a good chelator of copper and has been shown to inhibit tyrosinase from various sources including fungi, plants, and animal tissues [30].Kojic acid glucoside (2), isolated from plant sources, showed a good antiperspirant effect, but regarding antibacterial activity and tyrosinase inhibitory activity, kojic acid glucoside was less effective than kojic acid [31,32]. Galactosyl kojic acid (3), having a tyrosinase-inhibiting action and a melamine production-inhibiting action, is useful as an oxidation- and black coloration-preventing agent for foods and a whitening agent for cosmetics [33]. Kojic acid di-Me ether (4) and the known kojic acid monomethyl ether (5) have been isolated from the broth of the marine-derived fungus Alternaria sp. collected from the surface of the marine green alga Ulva pertusa. Each isolate was tested for its tyrosinase inhibitory activity. Metabolite (1) was found to have strong tyrosinase inhibitory activity, and other isolated compounds were less active [34].
Bikojic acid (6, also known as BGY-F) was found in cotton lint associated with aflatoxin contamination in cotton seed Gossypium hirsutum [35]. Bikojic acid could be produced by fungal endophytes. Several fungal endophytes, such as Alternaria tenuissima, Acremonium alternatum, Cladosporium cladosporioides, Chaetomium globosum, Drechslerella dactyloides (formerly Arthrobotrys dactyloides), Exserohilum rostratum, Epicoccum nigrum, and Paecilomyces sp., are known to be antagonists against plant pathogens, insects, and nematode pests and have been isolated from upland cotton (G. hirsutum) cultivated in the United States [36,37]. Metabolite 6-n-pentyl-a-pyrone (7) was found in the marine algicolous fungus of Myrothecium sp. [37]. The fungal strain (MFA58) was isolated from the surface of the marine green alga Enteromorpha compressa collected at Baegunpo (Busan). This metabolite (7) exhibited a tyrosinase inhibitory activity with an IC50 value of 6.6 µM, which is more active than the kojic acid (7.7 -M) currently being used as a functional personal care compound. An a-pyrone derivative, 6-[(E)- hept-1-enyl]-a-pyrone (8), with tyrosinase inhibitory activity was obtained from a marine isolate of the fungus Botrytis sp. Isolated compound exhibited tyrosinase inhibitory activity with an IC50 = 4.5 µM [38].
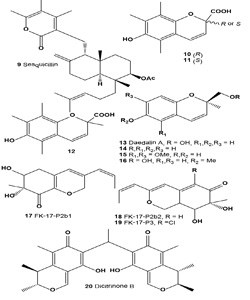
Sesquicillin (9) has been identified as an inhibitor of melanoma cell adhesion to the components of the extracellular matrix (ECM) in the cultured broth of fungal strain F60063. Sesquicillin strongly inhibited the adhesion of B16 melanoma cells to laminin, fibronectin, and type IV collagen [39]. Isolated compound was manufactured by aerobic fermentation of Sesquicillium globulisporum NRRL 5433, and/or the culture broth of Albophoma sp. FKI-1778 [40]. Sesquicillin has been identified as an inhibitor of melanoma cell adhesion to the components of the ECM in the cultured broth of fungal strain F60063. Sesquicillin strongly inhibited the adhesion of B16 melanoma cells to laminin, fibronectin, and type IV collagen. It also inhibited B16 melanoma cell invasion of reconstituted basement membrane Matrigel in vitro in a dose-dependent manner [39,40].
The inhibition of (10, R)-, (11, S)-, and (12) (±)-6-hydroxy- 2,5,7,8-tetramethylchroman-2-carboxylic acids (HTCCA) on mushroom tyrosinase was evaluated. All HTCCAs inhibited the tyrosinase activity. The ID50 values were 1.88, 1.84, and 1.88 µM for the (R)-, (S)-, and (±)-HTCCAs, respectively [41].
Daedalin A (13), (2R)-6-hydroxy-2-hydroxymethyl-2-methyl- 2H-chromene, was isolated from mycelial culture broth of fungi Daedalea dickinsii. Daedalin A strongly inhibited the activity of tyrosinase (IC50 = 194 µM) [42]. Bioactive metabolites (14-16) have showed tyrosine inhibitory activity [43].
UV-absorbing compounds FK17-P2a, FK-17-P2b1 (17), FK17- P2b2 (18), and FK17-P3 (19), useful for cosmetics, were manufactured by culturing Aspergillus versicolor FK-17 (FERM P-13621). The properties (including UV spectra) of the compounds and the properties of the A. versicolor were described. Azaphilone pigment FK17-P2b2 (18) was also isolated from the CH2Cl2 extract of a yellow mutant of the fungus Monascus kaoliang grown on rice [44].
Dicitrinone B (20), a rare carbon-bridged citrinin dimer, was isolated from the marine-derived fungus Penicillium citrinum. It was previously reported to have antitumor effects on tumor cells; however, the details of the mechanism remain unclear. Dicitrinone B inhibited the proliferation of multiple tumor types. Among them, the human malignant melanoma cell line A375 was confirmed to be the most sensitive [45].
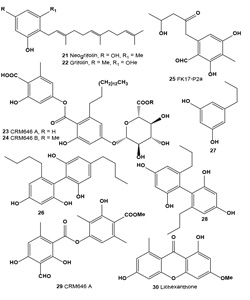
Aromatic and other metabolites: The mushroom Albatrellus confluens contains two active principles, neogrifolin (21) and grifolin (22), which are isomers with respect to the position of a farnesyl group on the aromatic ring. The tyrosinase inhibitory activity of (21) was 28% at 10 µM and 77% at 40 µM concentrations; the tyrosinase inhibitory activity of (22) was 18% at 500 µM and 64% at 900 µM concentrations [46]. The tyrosinase inhibitor neogrifolin was also isolated from the mushroom of Polyporus confluens, and at 50 ppm, it showed 100% tyrosinase inhibition [47].
CRM646 A (23) and B (24) were isolated as novel heparinase inhibitors from fungus AcremoniumMT70646 (family Hypocreaceae). Both compounds were active as melanoma metastasis inhibitors [48-50]. UV-absorbing metabolite FK17-P2a (25) was obtained from the fermentation broth of Aspergillus versicolor FK-17 (FERM P-13621) [44].
Compounds (26-28) were isolated from several Japanese lichens Alectoria nidulifera, Hypogymnia physodes, Nephromopsis ornate, Protousnea sp., and Umbilicaria kisovana. All compounds showed strong inhibition of melanin biosynthesis at low concentrations without cytotoxicity. It was also shown that melanin synthesis was inhibited in two ways, inhibiting both tyrosinase biosynthesis and tyrosinasecatalyzed reactions [51-53].
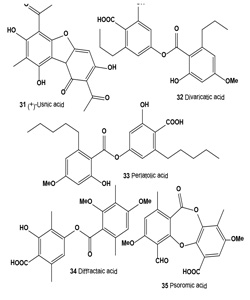
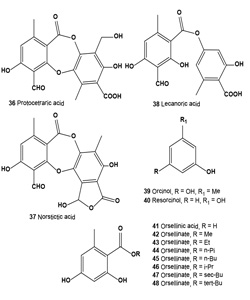
Atranorin (29), lichexanthone (30), and the (+)-usnic (31), divaricatic (32), perlatolic (33), diffractaic (34), psoromic (35), protocetraric (36), and norstictic (37) acids isolated from the lichens Parmotrema dilatatum, Usnea subcavata, Usnea sp., Ramalina sp., Cladina confusa, Dirinaria aspera, and Parmotrema lichexanthonicum. Tested compounds have showed activity against B16-F10 melanoma and UACC-62 cells and 3T3 normal cells.
A sulforhodamine B assay revealed significant cytotoxic activity in protocetraric, divaricatic, and perlatolic acids on UACC-62 cells (50% growth inhibitory concentration, GI50 = 0.5, 2.7, and 3.3 μg/mL, respectively). Divaricatic and perlatolic acids proved the most active on B16-F10 cells (GI50 4.4, and 18.0 μg/mL, respectively) and the most cytotoxic to 3T3 normal cells. Usnic (31), diffractaic (32), norstictic (37), and psoromic (35) acids were cytotoxic to UACC-62 cells in the 24.7 to 36.6 μg/mL range, as were protocetraric and diffractaic acids to B16-F10 cells (GI50 24.0, 25.4 μg/mL, respectively). Protocetraric acid (36) was highly selective (SI, selectivity index 93.3) against UACC-62 cells, followed by norstictic, perlatolic, psoromic, and divaricatic acids, whereas norstictic and divaricatic acids were more selective against B16-F10 cells. The high SI value obtained for protocetraric acid on UACC-62 cells makes it a potential candidate for the study of melanomas in experimental models. The action of lichen-derived compounds on many tumor cells has been a focus of evaluations for the last 3 decades [54].
Lecanoric acid (38), a lichen depside, was isolated from a Parmotrema tinctorum specimen and treated with alcohols to produce orsellinic acid (41) and orsellinates to 2,4-dihydroxy- 6-n-methyl benzoates. Free radical scavenging activity of methyl (42), ethyl (43), n-propyl (44), n-butyl (45), iso-propyl (46), sec-butyl (47), tert-butyl (48) orsellinates was evaluated using the 2,2-diphenyl-1-picrylhydrazyl (DPPH) method. The results showed that chain elongation of methyl (42) to n-butyl (45) causes a rise in the antioxidant activity. However, iso-propyl (46) and tert-butyl (48) were more active than the corresponding linear compounds, although sec-butyl (47) was less active among the chain-ramified compounds. All of the orsellinates were less active than lecanoric acid (38) and orsellinic acid (41). Orcinol (39) and resorcinol (40) were also studied for comparison with the activities of orsellinates [55-57].
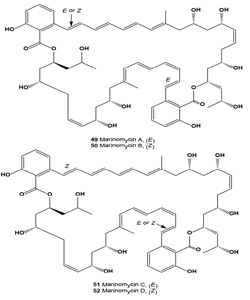
Four antitumor antibiotics of an unusual structure class, the marinomycins A-D (49-52, respectively), were isolated from the saline culture of marine actinomycetes Marinispora sp. [58]. In standard room light, marinomycin A (49) slowly isomerizes to its geometrical isomers marinomycins B and C. Marinomycins A-D all showed significant antimicrobial activities against drug resistant bacterial pathogens and demonstrated impressive and selective cancer cell cytotoxicities against 6 of the 8 melanoma cell lines in the National Cancer Institute's 60 cell line panel [58].
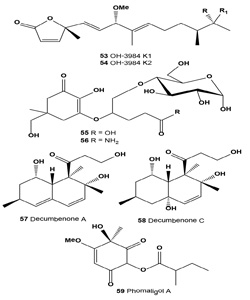
Melanogenesis inhibitors OH-3984 K1 (53) and K2 (54) were isolated from a fermentation broth of Streptomyces sp. OH- 3984. Both OH-3984 K1 and K2 inhibited the melanogenesis of B16 melanoma cells at concentrations of 7.5 and 3.8 μg/ mL, respectively [59].
It has been found that rock-inhabiting fungi also produce considerable amounts of mycosporine-glutaminol-glucoside, absorbing in the UV range with a wavelength maximum centered approximately 310 nm [60]. Initially, mycosporines were described as differentiative or reproductive markers in fungal spores and mycelia of Morchella esculenta [60,61]. At present, multiple cellular functions, such as osmoprotective, antioxidant, and UV-protectant roles, are assigned to these substances [24,57,62,63].
Microcolonial ascomycetes are known to inhabit bare rock surfaces in cold and hot deserts and thus they are habitually exposed to high levels of solar radiation [21,57]. Several of these stress-tolerant fungal isolates, cultivated under daylight illumination, were studied for the presence of effective UV radiation protecting substances. It was demonstrated that both mycosporine-glutamicol-glucoside (55) and mycosporine- glutaminol-glucoside (56) are natural secondary metabolites of microcolonial fungi [64].
Decumbenone A (57) was isolated from the fungus Penicillium decumbens as an inhibitor of melanin formation by the rice blast pathogen Magnaporthe grisea [65]. This compound was found in the extract of the marine-derived strain of the fungus Aspergillus sulphureus on the growth of seedling roots of buckwheat, wheat, barley, and corn [66]. A new decaline derivative, decumbenone C (58) along with four known compounds, decumbenones A (57) and B, diorcinol, and brevianamide F, were isolated from the marine fungus Aspergillus sulphureus KMM 4640. Decumbenone C shows potent cytotoxic activity against SK-MEL-5 human melanoma cells with an IC50 value of 0.9 µ? [67].
Tyrosinase inhibitor phomaligol A (59) has been isolated from the broth of the marine-derived fungus Altenaria sp. collected from the surface of the marine green alga Ulva pertusa [68].
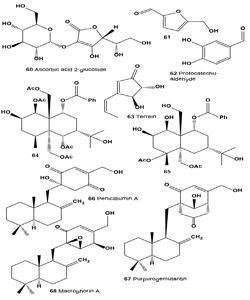
Ascorbic acid 2-glucoside (60) was present as an active ingredient in the extract of the pine mushroom Tricholoma matsutake. The compound has a dramatically increased inhibition effect on tyrosinase activity, exhibits a skin whitening effect, and has high stability in formulation [69].
Two tyrosinase inhibitory compounds, 5-hydro-xymethyl- 2-furaldehyde (61) and protocatechu-aldehyde (62), were isolated from the fruiting body of a medicinal mushroom Phellinus linteus [70].
Terrein (63), a bioactive fungal metabolite, was isolated for the first time from Aspergillus terreus in 1935 [71]. Terrein was first found to strongly decrease tyrosinase production by down-regulating MITF via ERK activation, and thus inhibiting melanin synthesis [72,73]. More recently, terrein was found to induce sustained ERK activation and MITF down-regulation, and luciferase assays showed that terrein inhibits MITF promoter activity in a dose-dependent manner. To elucidate the correlation between ERK pathway activation and a decreased MITF transcriptional level, PD98059, a specific inhibitor of the ERK pathway, was applied before terrein treatment and found to abrogate the terrein-induced MITF attenuation. These results suggest that terrein reduces melanin synthesis by reducing tyrosinase production via ERK activation and that this is followed by MITF down-regulation [74].
Two sesquiterpenes, 1β,5α,6α,14-tetraacetoxy-9a- benzoyloxy-7βH-eudesman-2β,11-diol (64) and 4a,5a- diacetoxy-9a-benzoyloxy-7βH-eudesman-1β,2β, 11,14-tetraol (65), were produced as stress metabolites in the cultured mycelia of fungus Pestalotiopsis sp. Z233 isolated from the brown marine algae Sargassum horneri in response to abiotic stress elicitation by CuCl2. Both compounds (64) and (65) showed tyrosinase inhibitory activities with IC50 values of 14.8 µM and 22.3 µM, respectively [75].
A fungal strain, Penicillium sp. F00120, was isolated from a deep sea sediment sample during a screening program and the EtOAc extract displayed strong toxic activity against brine shrimp larvae. One sesquiterpene, quinone penicilliumin A (66), and two known compounds, ergosterol and ergosterol peroxide, were isolated from the culture of solid-state fermentation of this strain. Penicilliumin A exhibited in vitro cytotoxic activity against B16, A375, and Hela cells [76].
Two drimenyl cyclohexenone derivatives, purpurogemutantidin (66), purpurogemutantin (67) and macrophorin A (68), were isolated from a bioactive mutant BD-1-6 obtained by random diethyl sulfate mutagenesis of a marine-derived Penicillium purpurogenum G59. Compounds (66) and (67) significantly inhibited human cancer K562, HL-60, HeLa, BGC-823, and MCF-7 cells, and compound (68) also inhibited the K562 and HL-60 cells [77].
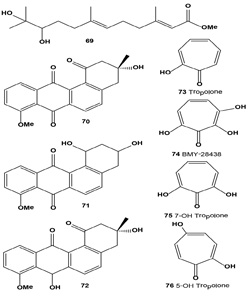
Melanin synthesis inhibitor (S)-(-)-10,11-dihydroxy-farnesoic acid methyl ester (69) was found in the culture broth from an insect morphopathogenic fungus, Beauveria bassiana CS1029 [78]. Furthermore, Baek and co-workers compared levels of TYR, TRP-1, TRP-2, MITF, and MC1R mRNA expression by reverse-transcription polymerase chain reaction and showed that treatment of Melan-A cells with 35 µM (69) led to an 11-fold decrease in TYR expression, a six-fold decrease in TRP- 2 expression, and a five-fold decrease in MITF expression. Together, these results indicate that (69) is a potent inhibitor of melanin synthesis that can potentially be used for cosmetic biomaterial(s) [78].
Streptomyces flavidofuscus cultivated in an arginine glycerol medium produced three major metabolites identified as (-)-8-O-methyltetrangomycin (70), 8-O-methyltetrangulol (71) and 8-O-methyl-7-deoxo-7-hydroxytetrangomycin (72). Isolated angucycline metabolites showed significant antimicrobial activity against Bacillus cereus and Listeria monocytogenes. Cytotoxic activities of compounds (70-72) against four cell lines, B16 melanoma cells, HT-29 and nontumor V79 and L929, were evaluated. Compound (72) was the most potent anticancer agent, with an IC50 of 0.054 µg/ mL against melanoma cell line B16 [79].
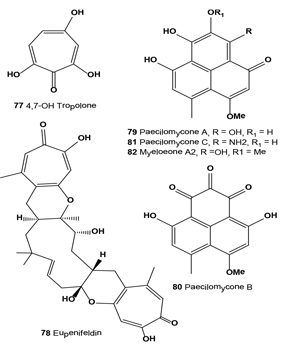
Tropolone (73) and its derivatives have been isolated for the first time from fungus Talaromyces stipitatus (or Penicillium stipitatum) and Penicillium puberulum, and more recently Dewar has reported a compound containing a non-benzenoid aromatic seven-membered ring (2-hydroxy-2,4,6-cycloheptatrienone), which he termed a tropolone. An antitumor antibiotic BMY-28438 (74) and other tropolone derivatives (75-77) were isolated from the cultured broth of Streptomyces tropolofaciens No K611-97 [80-82].
The antibiotic showed potent cytotoxicity against cultured B16 melanoma cells and a remarkable prolongation of lifespan in mice bearing B16 melanoma [83]. Eupenifeldin (78) was isolated from cultures of anamorph fungus Eupenicillium brefeldianum ATCC 74184 by extraction and crystallization. The compound was identified as a pentacyclic bistropolone and showed cytotoxic activities against several cancer cell lines [84].
By screening fifty strains of entomopathogenic fungi and rescreening seven strains of Paecilomyces gunnii, a methanol extract of liquid-cultivated mycelia of P. gunnii was found to have the strongest tyrosinase inhibitory activity. Compounds (79-82), with half inhibition concentrations IC50 of 0.11, 0.17, and 0.14 µM against diphenolase, respectively, were obtained from the extract. Structure and activity studies showed that tyrosinase inhibition activities are positively related to the number of hydroxyl groups on the paecilomycones [85].
Alkaloids and nitrogen-containing metabolites
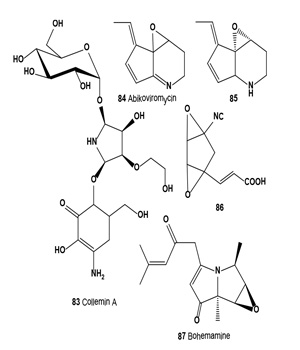
Lichens (lichenized ascomycetes and/or lichenized basidiomycetes) are unique symbiotic organisms comprising fungi and algae (or cyanobacteria); they produce n-alkanes, unusual betaine glycerols, arsenolipids, and unsaturated and halogenated fatty acids [20-25,86-98]. Many different bioactive secondary metabolites have been isolated from lichen species and subsequently used in pharmaceutical sciences [24,25,57]. Torres and co-workers isolated a photoprotective mycosporine, Collemin A (83), from cultivated lichenized ascomycete Collema cristatum[99,100].
Collemin A (83) prevented UV-B induced cell destruction in a dose-dependent manner and decreased pyrimidine dimer formation. Cell survival was estimated using the trypan blue exclusion assay [99]. In vivo biological activity of the isolated compound was assayed for the ability to prevent UV-B induced erythema (sunburn) of human skin. The pure compound was applied to the inside forearm of a volunteer 30 min prior to four MEDs (Minimal Erythema Dose) of UV-B irradiation.
Streptomyces strain SF-973, which produces antibiotics SF- 973 A, B, and C, was isolated from a soil sample collected in Hiroshima (Japan). SF-973 B catalyzed the conversion of inactive SF-973 C (dihydroabikoviromycin) to bioactive SF-973 A (abikoviromycin). Abikoviromycin (84) and dihydroabikoviromycin (85) are inhibitors of polyketide synthase (PKS1) involved in melanin biosynthesis by Colletotrichum lagenarium. Obtained results showed that PKS1 inhibitors are potential effective drug candidates for selective inhibition of melanin production of pathogenic fungi [101,102].
Anti-melanoma agent (86) was isolated from the fermentation broth of fungus Talaromyces FO-3182. This compound (86) reduced melanin content of B16 melanoma cells to 75% at a final concentration of 1-5 M without any significant effects on the cell number. The compound showed strong inhibitory activity of tyrosinase prepared from B16 melanoma cells with a concentration of 50% inhibition at approximately 5 nM [103].
Investigation of the culture extracts of a marine-derived Streptomyces sp. led to the isolation of bohemamine-type pyrrolizidine alkaloid, anticancer bohemamine B (87) [104]. An AMA bohemamine (87) was also isolated as a product of Actinosporangium species (C36145, ATCC 31127) [105].
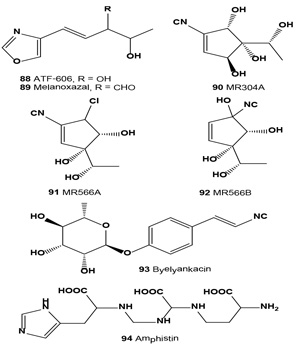
Low-toxicity melanin-biosynthesis inhibitor ATF-606 (88) was produced by culturing Trichoderma ATF-606 and Trichoderma sp. ATF-451. This compound inhibits melanin synthesis, using silkworm body fluid as an indicator, and tyrosinase of mushrooms [106]. Melanoxazal (89) also showed a strong inhibitory activity against mushroom tyrosinase with an IC50 = 4.2 µg/mL [107]. An isonitrile inhibitor of melanin synthesis, MR304A (90), was isolated from the culture broth of Trichoderma harzianum. Compound (90) inhibited melanin formation in Streptomyces bikiniensis and B16 melanoma cells and inhibited mushroom tyrosinase activity [108].
Melanin synthesis inhibitors (MR566A, 91 and B, 92) and six related known isocyanocyclopentenes were previously isolated from the fermentation broth of Trichoderma harzianum. The IC50 values of MR566A and B against mushroom tyrosine were 1.72 and 47 µM, respectively. They inhibited melanin biosynthesis in B16 melanoma cells with MIC values of 0.1 and 2.2 µM, respectively [109].
A melanogenesis inhibitor, byelyankacin (93) was obtained as a white powder from the culture medium. Byelyankacin potently inhibited mushroom tyrosinase and melanogenesis of B16-2D2 melanoma cells with IC50 values of 2.1 nM and 30 nM, respectively [110].
A melanogenesis inhibitor named amphistin (94) was isolated from the fermentation broth of an actinomycete strain KP-3052. Amphistin inhibited the melanogenesis of B16 melanoma cells at a concentration of 6.8 µM [54].
Several melanin production inhibitors, such as fusarin C (95), (7Z)-fusarin C (96) and (5Z)-fusarin C (97), were isolated from extracts of plant pathogen fungus Nectria coccinea. All compounds inhibited the formation of 1,8-dihydroxyaphthalene/melanin by Lachnellula sp. beginning at 1-2 µg/disk. Cytotoxic and phytotoxic activities were also reported [112].
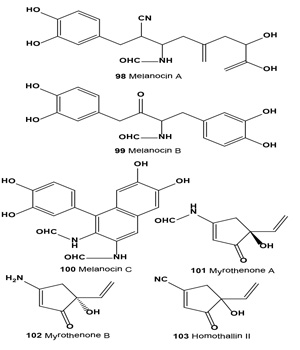
Melanin synthesis inhibitors melanocins A (98), B (99) and C (100) were isolated from the fermentation broth and extract of mycelium of Eupenicillium shearii F80695. Melanocin A, an isocyanide compound, inhibited mushroom tyrosinase and melanin biosynthesis of B16 melanoma cells with an IC50 value of 9.0 nM and MIC value of 0.9 µM [113].
The tyroinase inhibitors 3-amino-5-ethenylcyclo-pentenones, myrothenones A (101) and B (102), were isolated from the marine algicolous fungus of Myrothecium sp. [114]. The tyroinase inhibitor homothallin II (103) is produced by soil fungus Trichoderma viride strain H1-7 from a marine environment. Compound (103) was previously isolated as an antibiotic from Trichoderma koningii and T. harzianum. The inhibitor showed competitive inhibition against mushroom tyrosinase [115].
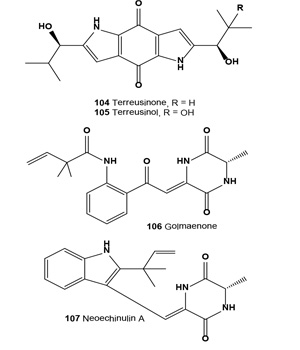
A chiral dipyrrolobenzoquinone derivative, terreusinone (104), has been isolated as a potent UV-A protectant from the marine algicolous fungus Aspergillus terreus. This compound exhibited UV-A absorbing activity with an ED50 value of 70 µg/mL [116]. Preparative-scale fermentation of terreusinone with a Streptomyces sp. resulted in the isolation of an oxidized metabolite, terreusinol (105). Isolated alkaloid showed UV-A (320-390 nm) protecting activity with an ED50 value of 150 µM, which is more active than oxybenzone (ED50, 350 µM), the current active ingredient in sunscreen [117].
A diketopiperazine alkaloid, golmaenone (106), and neoechinulin A (107) have been isolated from the culture broth of the marine-derived fungus Aspergillus sp. These alkaloids exhibited significant radical scavenging activity against 1,1-diphenyl-2-picrylhydrazyl with IC50 values of 20 and 24 µM, respectively, which were similar to the positive control, ascorbic acid (IC50, 20 µM). Compounds (106 and 107) also showed UV-A (320-390 nm) protecting activity with ED50 values of 90 and 170 µM, respectively, which are more active than oxybenzone (ED50, 350 µM) [118]. In addition, a radical scavenging and UV-A protecting prenylated diketopiperazine alkaloid, dihydroxy-isoechinulin A (108), was described from a marine isolate of the fungus Aspergillus sp. [119].
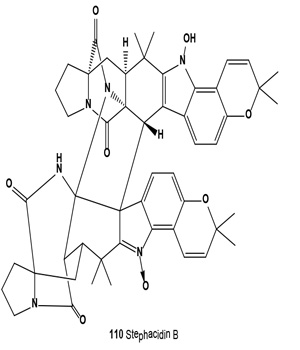
Avrainvillamide (109) was produced from a fermentation of the marine fungus Aspergillus CNC358. Avrainvillamide was demonstrated to be active against the human colon tumor cell line HCT-166 and showed significant selective in vitro cytotoxicity towards the melanoma cell line MALME-3M and the human breast cancer cell lines βT-49 and T-470 [120].
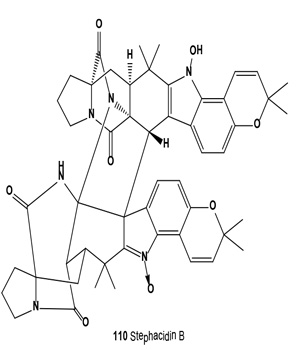
Antitumor antibiotics, designated stephacidin A and stephacidin B (110), were produced by fermentation of Aspergillus ochraceus ATCC-74432. These antibiotics inhibit the growth of mammalian tumors, particularly prostate carcinoma [121]. Both antibiotics were also isolated from the solid fermentation of Aspergillus ochraceus WC76466. Both alkaloids exhibited in vitro cytotoxicity against a number of human tumor cell lines, but stephacidin B demonstrated more potent and selective antitumor activities, especially against prostate testosterone-dependent LNCaP cells, with an IC50 value of 60 nM [122]. Both avrainvillamide (109) and related dimer stephacidin B (110) have been reported to exhibit antiproliferative activity, whereas avrainvillamide has also been reported to exhibit antimicrobial activity against multi-drug resistant bacteria. Avrainvillamide has been found to target cytoskeleton-linking membrane protein (CLIMP-63), thereby preventing cells from undergoing mitosis. The antitumor activity of (+)-stephacidin B was detected as: GI50 = 550 nM vs. BT-549 human breast carcinoma; GI50 = 942 nM vs. T-47D human breast ductal carcinoma; GI50 = 987 nM vs. MALME-3M human melanoma; GI50 = 952 nM vs. LnCAP human prostate cancer [123].
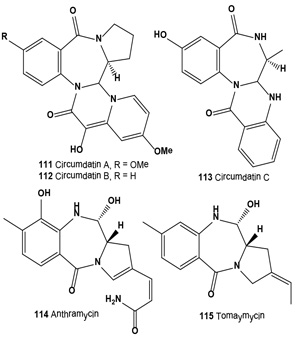
Three ultraviolet-A protecting benzodiazepine alkaloids, circumdatin A (111), B (112), and C (113), have been isolated from a terrestrial isolate of the fungus Aspergillus ochraceus [124]. More recently, these alkaloids have been isolated from the mycelium of a marine-derived anamorphic fungus of the genus Exophiala [125].
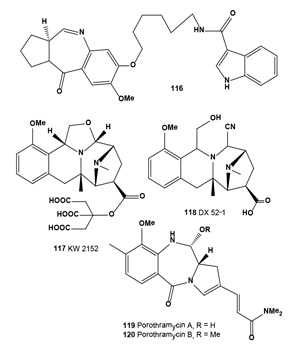
Anthramycin group antitumor antibiotics (114 and 115), isolated from a Streptomyces culture, showed activity against experimental tumors such as lymphocytic leukemia P388 ascites sarcoma-180, hepatoma AH130, Walker carcinosarcoma-256 and mouse mammary adeno-carcinoma [126,127]. Both antibiotics (114 and 115) showed in vitro cytotoxic activity against B16 melanoma cells. Antibiotic DC-81 (118) was isolated from cultures of Streptomyces roseiscleroticus DC-81 [128]. DC-81 is a PBD antitumor antibiotic produced by Streptomyces species. A derivative of DC-81 with an indole carboxylate moiety (116) had much higher sequence selectivity in DNA interactivity. An XTT assay demonstrated that the DC-81-indole conjugate possessed cytotoxicity against human melanoma cell lines [129].
Quinocarmycin monocitrate (117) and its analog, DX-52- 1 (118), demonstrated specificity for melanomas in the National Cancer Institute (NCI) in vitro human tumor cell line drug screen. In contrast to most cell lines, a 50% reduction in tumor cell burden at the end of a 48-h drug incubation was produced in five of eight melanoma lines by KW2152 concentrations (LC50) ranging from 0.5 to 10.9 µM and by DX- 52-1 concentrations ranging from 0.7 to 7.3 µM. DX-52-1 has been selected by the NCI for development in clinical trials, especially against melanomas. This agent represents one of the first to be selected for preclinical development based on disease-panel specificity discovered using the National Cancer Institute cancer drug screen [130]. The hydrocyanated derivative of the antitumor antibiotic quinocarmycin, DX-52-1 (118) was isolated from the culture broths of Streptomyces melanovinaceus and exhibits impressive activity against human melanoma xenograft models in vivo[131-133].
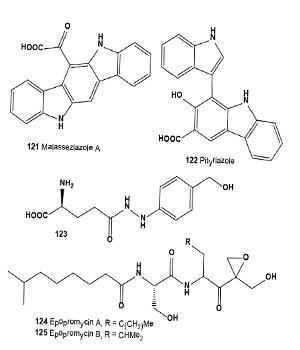
Antitumor antibiotics porothramycin A (119) and B (120) were produced by a strain of Streptomyces albus. The antibiotic was isolated in two active forms, the natural free hydroxyl form (porothramycin A) or the crystalline methyl ether form (porothramycin B) depending upon the isolation process used. The antibiotic exhibited antimicrobial activity against Gram-positive bacteria and anaerobes and significantly prolonged the survival times of mice implanted with experimental tumors from cell lines P388, L120 leukemia, and B16 melanoma [134]. An invention was described utilizing the anti-melanoma agents (121 and 122) produced by Malassezia with phenol oxidase inhibitory activity, which could be used as agents to treat hyper-pigmentation, malignant and semimalignant melanocytes and to inhibit their proliferation. The human pathogenic yeast Malassezia furfur was grown on tryptophan-rich medium. The indole derivatives can be used in topical formulations in combination with other active substances, e.g., antioxidants, sunscreens, and vitamins [135,136].
MPI agaritine, -[N-[?-(+)-L-glutamyl]-4-hydroxymethylphenyl]- hydrazine (123), was isolated and purified from the mushroom Agaricus bisporus [137]. This compound is characteristic for the genus Agaricus. Agaritine is a Chinese herbal medicine known for having antiviral and anticancer functions and for its potential as an anti-HIV agent [138].
Streptomyces NK04000 was thus found to produce the epopromycins A and B (124 and 125, respectively). Both epopromycins exhibit potent cytotoxicity against B16 melanoma (IC50 = 0.003 µg/mL) in vitro [139].
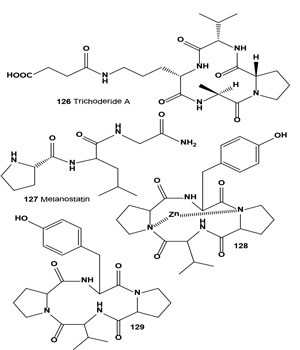
A cyclotetrapeptide, trichoderide A (126), was isolated from the mesophilic and filamentous fungus Trichoderma reesei. Trichoderide A showed moderate cytotoxicity against human A375-S2 melanoma cell line [140].
Melanostatin (127), an antibiotic with melanin synthesis inhibitory activity, was isolated from the fermentation broth of Streptomyces claviver No N924-2 [141]. Melanostatin strongly inhibited melanin formation in Streptomyces bikiniensis NRRL B-1049 and B16 melanoma cells. Lactobacillus helveticus JCM-1120 produced a melanin formation inhibitor, tetracyclopeptide (128, Zn-complex), which inhibited tyrosinase and was stable at 37° for =4 weeks in liquid condition [142]. The same cyclotetrapeptide cyclo(L-Pro-L-Tyr-L-Pro-L-Val) (129) was isolated from the same L. helveticus. Compound (129) was a strong inhibitor of mushroom tyrosinase, with an IC50 = 1.5 µM [143].
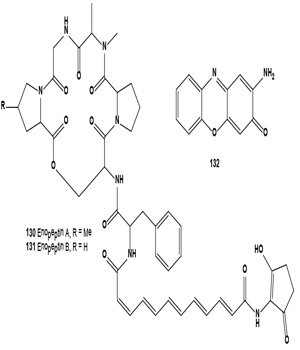
Using the Streptomyces sp. X59 as a melanin formation inhibitor screening model, two positive compounds, H7264 A and B, were isolated from the culture broth of Streptomyces sp. SIIA-H7264 collected from a soil sample in Yibin, Sichuan province, China. H7264 A and B were identical to melanin biosynthesis inhibitors Enopeptin A (130) and B (131), depsipeptide antibiotics with antiviral activity [144]. Melanogenesis inhibitor 2-amino-3H-phenoxazin-3-one (132) was identified from the mushroom Agaricus bisporus [144].
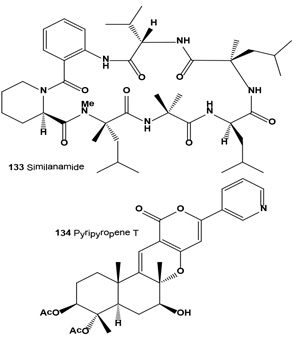
Cyclohexapeptide, similanamide (133), and a pyripyropene derivative named pyripyropene T (134) were isolated from the ethyl acetate extract of the culture of the marine spongeassociated fungus Aspergillus similanensis KUFA 0013. Isolated compounds exhibited weak in vitro growth inhibitory activity against MCF-7 (breast adenocarcinoma), NCI-H460 (non-small cell lung cancer) and A373 (melanoma) cell lines, as well as antibacterial activity against reference strains and the environmental multidrug-resistant isolates [145].
CONCLUSION
Melanin and tyrosinase inhibitors comprise a small group of natural products. They are primarily present in fungal species and have also been detected in plants and some invertebrates. Little information is known about the biological activities of these metabolites. Nevertheless, reported activities for these isolated compounds have shown strong melanin and tyrosinase inhibition, as well as anticancer, antibacterial, antiviral, and other activities. Referred metabolites have been shown to be promising candidates for the development of new drugs used for the treatment of the diseases indicated above.
Conflict of Interest The author confirms that this article content has no conflict of interest.
REFERENCES
-
Levitsky DO and Dembitsky VM. (2015). Anti-breast cancer agents derived from plants. Nat. Prod. Bioprospect. 5(1), 1-16.
-
Bens G. (2014). Sunscreens. Adv. Exp. Med. Biol. 810, 429- 463.
-
Yabuta T. (1922). Kojic by many species of Aspergillus, Acetobacter, and Penicillium. J. Chem. Soc. 122, 939-952.
-
Yabuta T. (1924). The constitution of kojic acid, a gamma-pyrone derivative formed by Aspergillus oryzae from carbohydrates. J. Chem. Soc. 125, 575-587.
-
Honda S. Antiperspirants containing kojic acids, Japanese Kokai Tokkyo Koho, 7 Japanese Patent 2,000,016,928, 8 June 2000.
-
Arai T and Sano H. (1994). Novel UV - absorbing compounds FK17-P2a, FK-17-P2b1, FK17-P2b2, and FK17-P3 and manufacture of the compounds with Aspergillus sp. Jpn. Kokai Tokkyo Koho, 6 Japanese patent: JP 06329576 19941129 Heisei, March 11.
-
Minosasa J, Matsui K, Uehara H and Tanaka H. (1991). Tyrosinase inhibitors containing neogrifolin. Jpn. Kokai Tokkyo Koho, 15 Japanese Patent: JP 03109319 A 19910509 Heisei.
-
Kinoshita Y. (1997). Pharmaceutical activities found in lichens. Techno-Cosmos. 11, 25-29.
-
Dembitsky VM and Tolstikov GA. (2005). Organic Metabolites of Lichens. Geo-Press, Novosibirsk (Russia), 284.
-
Gorbushina AA, Whitehead K, Dornieden T, Niesse A, et al. (2003). Black fungal colonies as units of survival: hyphal mycosporines synthesized by rock-dwelling microcolonial fungi. Can. J. Botany. 81(2), 131-138.
-
Yoo YG, Lee GT, Jung JH and Cho BG. (2004). Cosmetic composition for skin-whitening comprising ascorbic acid 2-glucoside and tricholoma matsutake extracts as active ingredients. Assignee Coreana Cosmetics. Korean Patent: 1020040038438.
-
Dewar MJ. (1945). Structure of stipitatic acid. Nature. 155, 50-51.
-
Torres A, Dor I, Rottem E, Srebnik M, et al. (2003). n-Alkane and fatty acid variations in the lichen Xanthoria parietina, their photobiont Trebouxia sp. and mycobiont, from Jerusalem hills. Eur. J. Biochem. 270, 2120-2125.
-
Dembitsky VM and Levitsky DO. (2004). Arsenolipids. Prog. Lipid Res. 43(5), 403-448.
-
Dembitsky VM and Levitsky DO. (2006). Acetylenic terrestrial anticancer agents. Nat. Prod. Commun. 1, 405-429.
-
Tanaka N, Naganuma M, Fukuda M, Wati Y, et al. (1996). Novel inhibitor of melanogenesis produced by Talaromyces FO-3182. Nippon Koshohin Kagakkaishi. 20(1), 3-6.
-
Nakagawa A and Takahashi S. (1996). Melanin-biosynthesis inhibitor ATF-606 manufacture with Trichoderma. Jpn. Kokai Tokkyo Koho, 8 pp. Japanese Patent: JKXXAF JP 08104695 A 19960423 Heisei. Application: JP 94-241092 19941005.
-
Lee SM, Li XF, Jiang H, Cheng JG, et al. (2003). Terreusinone, a novel UV-A protecting dipyrrolo-quinone from the marine algicolous fungus Aspergillus terreus. Tetrahedron Lett. 44(42), 7707-7710.
-
Fenical W, Jensen PR and Cheng XC. (2000). Avrainvillamide, a cytotoxic marine natural product, and the derivatives thereof. 6 US Patent: 6066635 A 20000523.>
-
Cutrone JQ, Krampitz KD, Shu Y-Z, Chang, et al. (2001). Stephacidin antitumor antibiotics. 15 pp. U.S. Ser. No. 329,151, abandoned. US Patent: 6291461 B1 20010918.
-
Myers AG, Herzon SB, WulffJE, Siegrist R, et al. (2006). Synthesis of avrainvillamide, stephacidin B, and analogs thereof as medicinals. PCT Int. Appl. 232. WO 2006102097 A2 20060928.
-
Zhang D, YangX, Kang JS, Choi HD, et al. (2008). Circumdatin I, a new ultraviolet-A protecting benzodiazepine alkaloid from a marine isolate of the fungus Exophiala. J. Antibiot. (Tokyo). 61(1), 40-42.
-
Anon. (1983). Antibiotic DC-81. Jpn. Kokai Tokkyo Koho, 9 Japanese Patent: JP 58180487 A 19831021 Showa.
-
Mayser P, Steglich W, Kraemer H-J and Irlinger B. (2002). Indole derivatives from Malassezia yeast with inhibitory effect on phenol oxidase. 6 German Patent: DE 10109005 A1 20020905.<
-
Irlinger B, Bartsch A, Kraemer H-J, Mayser P, et al.(2005). New tryptophan metabolites from cultures of the lipophilic yeast Malassezia furfur. Helv. Chim. Acta, 88, 1472-1485.
-
Somoto A and Kuranari A. (1993). Tetracyclopeptides as melanin formation inhibitors. Jpn. Kokai Tokkyo Koho, 10 pp. Japanese Patent: JP 05194258 A 19930803.
-
Lu Q, Tian M, Liu Y and Yu D. (2002). Isolation and structure elucidation of melanin biosynthesis inhibitors H7264 A and B. Zhongguo Kangshengsu Zazhi. 27(7), 385-386.