Previous Issues Volume 1, Issue 2 - 2016
Anatomical Modifications for Bipedal Locomotion in Modern Humans: A Mini Review
Ahmed Al-Imam*1,2
1Novel Psychoactive substances unit, University of Hertfordshire Doctoral College, Hertfordshire University, UK. 2Department of Anatomy and Cellular Biology, college of Medicine, University of Baghdad, Iraq.
Corresponding Author: Ahmed Al-Imam, Novel Psychoactive substances unit, University of Hertfordshire Doctoral College, Hertfordshire University, UK, Tel: +44 (0) 7482 571679; E-Mail: [email protected]
Received Date: 30 Mar 2017 Accepted Date: 31 May 2017 Published Date: 31 May 2017
Copyright © 2017 Al-Imam A
Citation: Al-Imam A. (2017). Anatomical Modifications for Bipedal Locomotion in Modern Humans: A Mini Review. Mathews J Orthop. 2(1): 016.
ABSTRACT
Several attributes are especial for primates, some of these are even more unique in humans. These may not be limited to; energy-saving bipedal posture and locomotion, complex manual skills and tool making abilities, social and cultural organisation, augmented cranial capacity, a more developed frontal lobe, and language proficiency. From a Darwinian point of view, erect posture was achieved to free the hands for tool-making. However, more recent theories suggested that bipedal locomotion was related to environmental factors which led to the advent of distinctive and remarkable anatomical features for a form of locomotion that is more energy-economic than quadrupedal locomotion. The purpose of this mini-review is to elaborate on the critical biomechanical and anatomical adaptations in modern human which enabled an energy-efficient form of bipedalism; this form of locomotion is never seen in other species including primates. These anatomic features and adaptations include; adjustments to the general body architectural plan, in addition to cranial and postcranial modifications. The latter is debated to be the most critical for a stable and fuel-efficient upright walking, other anatomic adaptation were parallel and complimentary. The most significant of these anatomical changes took place post-cranially at the level of the pelvis. Some of these changes can be found in Australopithecus afarensis dating back to 3.6 million years ago.
INTRODUCTION
Several features are unique to humans including; an efficient bipedal or upright walking, stable supine posture, an augmented cranial capacity and a complex frontal lobe, ability to speak and interpret speech, complex tool-making abilities, an elaborate social structure [1]. Cultural anthropologists and physical anthropologists consider that all these features contributed eventually to the development of a real culture, rather than a primitive culture (proto-culture) as seen in several primates species including chimpanzees [2-5]. It is estimated that the earliest of these traits to be established was the upright posture (bipedalism) in Australopithecine; including Lucy’s specie (Australopithecus afarensis) [6-9]. Numerous anatomic adaptations materialised independently across time; these changes exist cranially and post-cranially, principally serving the purpose of high energy efficiency. Compared to apes and quadrupeds, humans spend much less en ergy in bipedal locomotion [10]. Humans have also acquired longer lower limbs to serve the propulsive function during walking. Besides, humans have less body weight (BW) above the waist (68% of total BW in genus Homo vs. 82% in Apes) [11]. Additionally, the line of gravity passes behind the ears and slightly anterior to the spine, and anterior to the knees [10, 11]. Bipedalism appears to be unique and more advanced in humans, and particularly in genus Homo, than in all others primates. Musculoskeletal and anatomical adaptations can be specifically categorised into; general (1), post-cranial (2), and cranial (3). The changes started to be perfected in Australopithecus afarensis at approximately 3.6 million years; it has been extensively studied via the Laetoli footprints discovered by Mary Leaky in 1978 near Olduvai gorge in Tanzania [12, 13]. On the other that hominids adaptations, particularly pelvic redesigning, were far different for example that of Oreopithecus bambolii [14]. It is important to bring the attention of those who read this manuscript that the purpose of this review is not to take sides neither with the theory ofDarwinian’s evolution nor the theory of intelligent design. The renowned Darwinian evolutions by Charles Darwin and Alfred Russel claims that the organic lifeforms are in a constant and a slowly progressive evolution across vast aeons of time, and all species had common ancestry [15-17]. On the other hand, other scholars claim the theory of an intelligent design of life and humans to be specific, and that randomness is not destined to create an intelligent and an aware creature like the modern humans [18-20].
General Modifications Bipedal walking in humans is estimated to be 75% less energy-costing than both quadrupedal and bipedal locomotion in chimpanzees while running in humans is about 75% less efficient than walking also in humans [7, 21, 22]. There was a change in lower limb length and the leg length specifically, the purpose is to provide an efficient lever mechanism in pushing (propelling) the body forward, and to reduce the need for the muscular efforts during the swinging phase pf upright walking [23-25]. On the other hand, the upper limbs were excluded from locomotions in humans, except in infancy. Hence, the upper limbs, particularly the hands, became optimised to carry and manipulate objects with high manual precisions. The implications of this change in motor function had led to a reduction in the ratio of humerus-to-femur length. Humans have a different distribution of weight above and below the level of the waist; it is estimated the about 68% of the total body weight in humans (versus 82% in apes) is located above the waist [11]. Furthermore, the line of gravity is located slightly anterior to the vertebral columns and the knees [10, 11, 26].
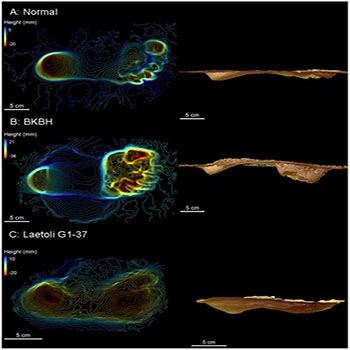
Post-Cranial Adaptations Post-cranial adaptations were of paramount importance for a coherent and a cost-effective bipedal form of locomotion. The foot has evolved to function as the propulsive organ and of no grasping functionality; the heal became enlarged and positioned beneath the centre of gravity, the toes were shorter and more straight (not for grasping), the hallux became fullyadducted and non-apposable [27, 28]. It can be deduced from the Laetoli footprints (Figure 1) of Australopithecus afarensis that the body weight was primarily transmitted down via the line of gravity to; the heel, the ball of the foot, the lateral foot border, and the big toe (hallux). Though the foot became arched, with two longitudinal arches and one transverse arch, it became more rigid when compared to monkeys, apes, and non-human primates [13, 27, 28]. Numerous pathologies may occur in the foot including, osteoarthritis, pes planus (flat foot), and hallux valgus; all these can interfere with the critical biomechanics involved in walking [29].
Figure1: Three-dimensional scans of experimental footprints (a, b) and a Laetoli footprint (c) (Raichlen et al., 2010).
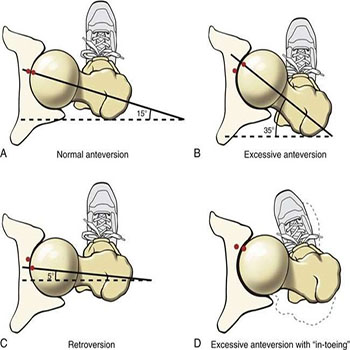
The knee became bigger, with a larger surface area (SA) of femoral-tibial condyles. Compared to apes, the condyles are more flattened and of longer anteroposterior (AP) diameter [30, 10]. Furthermore, the lateral femoral condyle possessed a unique lip to prevent lateral displacement (dislocation) of the patella from the patellofemoral unit; this lateral displacement occurs due to the pulling effect quadriceps femoris muscle in the presence of a valgus angle of the knee. The knees were allowed to be fully extended during the swinging phase of walking which potentiates propulsion from the ground. The knees developed a valgus angle (Q angle), thus positioning the knees right underneath the centre of gravity, which also enabled the knee to be locked in full extension, which can be maintained for an extended period of time while requiring a minimal muscular effort [31]. At the hip, the surface area of the coxo-femoral (hip) joint became increased, while the femur developed a conspicuous angle of torsion-inclination (Figure 2) including the collodiaphyseal angle to accommodate tensile, compressive, and torsional forces across the femoral neck and head [30, 32]. The cortex of the femoral neck became more thickened inferiorly; its trabecular pattern was rearranged to prevent and reduce the incidence of catastrophic fractures of the femoral neck [33]. The iliopsoas, the most powerful flexor muscle of the hip, became more de-veloped and an imprint was created in front the coxal bone immediately lateral to the iliopubic eminence [30].
Figure 2: The angle of torsion of femur: normal condition (a) and pathological conditions (b-d) (Musculoskeletal Key, 2016).
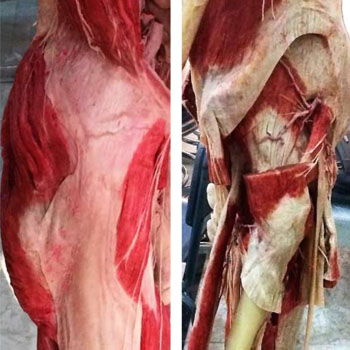
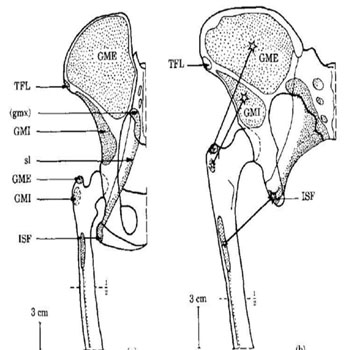
At the pelvis, there was an enlargement of the sacroiliac joints connecting the axial to the peripheral skeleton, both joints (bilaterally) became in a more precise alignment with; the acetabulum of the innominate bone (1), and the line of gravity (2). The ilia became modified in shape from long and narrow to short and broad. Additionally, there was broadening of the pelvis, the sacrum became broader with the sacral ala expanded more laterally, the sacrum also became more inclined backwards in continuity with lumbar lordosis, thus the volume of the pelvic cavity became significantly larger, these changes are more evidently noticed in females in an aim to accommodate the presenting part of the newborn baby at the time of delivery [34]. The ischial spines became more prominent medially providing robust attachments for the sacrospinous and sacrotuberous ligaments, which lead to the formation of a basin-like support for abdominal viscera; this basin-like support relies on the thoracic cage in quadrupeds [35, 34]. One of the most significant changes in the pelvis took place at iliac blades (Figure 3); the blades rotated axially, thus repositioning the anterior glutei muscles (gluteus medius and gluteus minimus) more anteriorly, while the gluteus maximus (the most powerful hip extensor muscle) was fixed posteriorly to persist as an extensor muscle of the coxo-femoral joint, thus preventing the body from pitching forward. The new post of anterior gluteal muscles (Figure 3 and 4) enabled them to function as the main abductor muscles of the hip joint, which led to an efficient and an accurate tilting of the pelvis during walking. Finally, the anterior iliac spines became robust, for anchorageof sartoriusand rectus femoris, which enabled these two muscles to function as stronger flexors of the thigh [6].
Figure 3: Superficial dissection of right lower limb (3a, left image) showing gluteus maximus, iliotibial tract, and tensor fascia lata. Deep dissection of left lower limb (3b, right image) showing the anterior glutei; gluteus medius and gluteus minimus, both muscles are abductors of the thigh in humans.
Figure 4: Gluteal muscles arrangement around the coxofemoral joint in a chimpanzee (a) and an Australopithecus afarensis (b) (Berge, 1994).
The vertebral column developed four curvatures in the sagittal plane; cervical, thoracic, lumbar, and sacral. It was Leonardo da Vinci who had 1st studied the double-Sigmoid curvature of the spinal column [36-38]. The aim of this double-S curvature of the spine is to withstand compressive forces more efficiently and to function as a shock absorber. The combined lumbar lordosis and thoracic kyphosis have positioned the centre of gravity directly above feet, preventing the body from toppling forward during walking. Moreover, the size of vertebrae, particularly the vertebral bodies and their facet joints, was significantly increased as we go down the spinal column till reaching the 1st piece of the sacrum (Nakatsukasa, Hayama, and Preuschoft, [39-41].
Cranial Adaptations These adaptations were significantly less numerous and less vital when compared with post-cranial adaptation. The head has become well positioned and accurately balanced on top of the cervical segment of the vertebral column; this has occurred due to the central positioning of the foramen magnum in relation to the anterior-posterior (AP) diameter of the cranium. Furthermore, the face has become orthognathic rather than prognathic as in non-human primates and Australpethicines. There was also a reduction in the amount of muscular mass of neck extensors. Similarly,there was a reduction of the supra-orbital ridges and its functioning occipitofrontalis muscle [30]. The endocranial capacity has significantly increased up to 1500 cubic centimeters over the period from 2.4 to 0.5 million years ago million year ago (Mya), this has also led to a change in the weight distribution within the cranium around the pivotal atlanto-occipital joint [42-45].
CONCLUSION
The upright walking, also known as bipedalism, is not strict feature for humans. However, a stable bipedal stance and an energy-economic bipedal form of locomotion are unique in humans compared with the large apes and non-human primates. It is a shared biomechanical trait between several different species of human and non-human primates. However, bipedal upright walking seems to be far evolved and more crafted in an advanced way in genus Homo, particularly in modern humans. The erect bipedal posture and locomotion were not easily achieved by humans; the evolutionary timeline has struggled to intelligently design a bold stance, and engineer an energy-efficient bipedalism. Nevertheless, this economic characteristic of locomotion was also accompanied by several pathologies affecting multiple elements of the musculoskeletal system including; the vertebral column, the hip joint, and the knee joints. Arthritis is one of the most devastating conditions affecting the musculoskeletal system, which simply occur as a consequence of the ageing process and excessive use of the joints. In this mini-review, the anatomical and biomechanical properties of human adaptations for bipedal walking were explored; these included cranial and postcranial adaptations and some other miscellaneous adaptation. It is apparent that adaptations at the level of the spine, pelvis,hip, andknee joint were the most critical for a successful bipedal locomotion. Each of these adaptations possibly occurred independently from the other and in a non-simultaneous fashion across aeons of the evolutionary timeline. Perhaps, bipedalism is the oldest of the unique traits in humans; other adaptations include; advanced tool-making abilities, progressively enlarged frontal lobe capacity, culture, verbal and comprehensive linguistic abilities, and written language.
ACKNOWLEDGMENTS
The author would like to acknowledge with gratitude the supportive efforts by Professor Dr Ashok Sahai. Professor Sahai is an eminent anatomist from an India; he is also a fellow and the vice president of the Anatomical Society of India (ASI). The author is grateful for Raichlen DA and Berge C, for their invaluable biomechanical studies which has been used to a large extent in the citation of this manuscript.
REFERENCES
- Matsuzawa T, Waal FB and Ferrari PF. (2012). What is uniquely human? A view from comparative cognitive development in humans and chimpanzees. The primate mind. 2: 288-305.
- Daly GB. (2015). Why Japanese primatology? A perspective from sociocultural anthropology. InPrimate Research Supplement The 31th Congress Primate Society of Japan. 106-106.
- Gamkrelidze TV and Ivanov VV. (1995). Indo-European and the Indo-Europeans: A Reconstruction and Historical Analysis of a Proto-Language and Proto-Culture. Part I: The Text. Part II: Bibliography, Indexes. Walter de Gruyter.
- McGrew WC. (2015). The cultured chimpanzee: nonsense or breakthrough. Human Ethology Bulletin. 30(1): 40-51.
- Williams N. (2008). Japan wildlife boost. Current Biology. 18(9): R358-R359.
- Berge C. (1994). How did the australopithecines walk? A biomechanical study of the hip and thigh of Australopithecus afarensis. Journal of Human Evolution. 26(4): 259-273.
- Sellers WI, Cain GM, Wang W and Crompton RH. (2005). Stride lengths, speed and energy costs in walking of Australopithecus afarensis: using evolutionary robotics to predict locomotion of early human ancestors. Journal of the Royal Society Interface. 2(5): 431-441.
- Stern JT and Susman RL. (1983).The locomotor anatomy of Australopithecus afarensis. American Journal of Physical Anthropology. 60(3): 279-317.
- Ward CV. (2013). Postural and locomotor adaptations of Australopithecus species. The paleobiology of Australopithecus. 235-245.
- Morbeck ME. (1986). Primate morphophysiology, locomotor analyses and human bipedalism. 423- 425.
- Rodman PS and McHenry HM. (1980). Bioenergetics and the origin of hominid bipedalism. American Journal of Physical Anthropology. 52(1): 103-106.
- Hatala KG, Demes B and Richmond BG. (2016). Laetoli footprints reveal bipedal gait biomechanics different from those of modern humans and chimpanzees. The Royal Society. 283(1836): 20160235.
- Raichlen DA, Gordon AD, Harcourt-Smith WE, Foster AD. et al. (2010). Laetoli footprints preserve earliest direct evidence of human-like bipedal biomechanics. PLoS One 5(3): e9769.
- . Köhler M and Moya-Sola S. (1997). Ape-like or hominidlike? The positional behavior of Oreopithecus bambolii reconsidered. Proceedings of the National Academy of Sciences. 94(21): 11747-11750
- Gould SJ. (1987). Is a new and general theory of evolution emerging?. InSelf-Organizing Systems. 113-130.
- Huxley TH. (1968). On the origin of species. University of Michigan P.
- Smith JM. (1993).The theory of evolution. Cambridge University Press.
- Dembski WA and McDowell S. (2008). Understanding intelligent design. Harvest House Publishers.
- Dembski WA. (2002). Intelligent design: The bridge between science & theology. InterVarsity Press.
- Pennock RT. (2001). Intelligent design creationism and its critics: Philosophical, theological, and scientific perspectives. MIT Press.
- Sellers WI, Dennis LA and Crompton RH. (2003). Predicting the metabolic energy costs of bipedalism using evolutionary robotics. Journal of Experimental Biology. 206(7): 1127- 1136.
- Wheeler PE. (1991).The influence of bipedalism on the energy and water budgets of early hominids. Journal of Human Evolution. 21(2): 117-36.
- Gatesy SM and Biewener AA. (1991). Bipedal locomotion: effects of speed, size and limb posture in birds and humans. Journal of Zoology. 224(1): 127-147.
- Steudel-Numbers KL and Tilkens MJ. (2004).The effect of lower limb length on the energetic cost of locomotion: implications for fossil hominins. Journal of Human Evolution. 47(1-2): 95-109.
- Steudel-Numbers KL. (2006). Energetics in Homo erectus and other early hominins: the consequences of increased lower-limb length. Journal of Human Evolution. 51(5): 445-453.
- Le Huec JC, Saddiki R, Franke J, Rigal J, et al. (2011). Equilibrium of the human body and the gravity line: the basics. European Spine Journal. 20(5): 558.
- Harcourt-Smith WE and Aiello LC. (2004). Fossils, feet and the evolution of human bipedal locomotion. Journal of Anatomy. 204(5): 403-416.
- Latimer B and Lovejoy CO. (1989). The calcaneus of Australopithecus afarensis and its implications for the evolution of bipedality. American Journal of Physical Anthropology. 78(3): 369-386.
- Wafai L, Zayegh A, Woulfe J, Aziz SM, et al. (2015). Identification of foot pathologies based on plantar pressure asymmetry. Sensors. 15(8): 20392-20408.
- Crompton RH, Sellers WI and Thorpe SK. (2010). Arboreality, terrestriality and bipedalism. Philosophical Transactions of the Royal Society B: Biological Sciences. 365(1556): 3301-3314.
- Mizuno Y, Kumagai M, Mattessich SM, Elias JJ, et al. (2001). Q-angle influences tibiofemoral and patellofemoral kinematics. Journal of Orthopaedic Research. 19(5): 834-840.
- Musculoskeletal Key. (2016). Structure and Function of the Hip.
- Brown TD and Ferguson AB. (1980). Mechanical property distributions in the cancellous bone of the human proximal femur. Acta Orthopaedica Scandinavica. 51(1-6): 429-437.
- Snell CA and Donhuysen HW. (1968).The pelvis in the bipedalism of primates. American journal of physical anthropology. 28(3): 239-246.
- Berge C. (1998). Heterochronic processesin human evolution: an ontogenetic analysis of the hominid pelvis. American Journal of Physical Anthropology. 105(4): 441-459.
- Crisco Iii JJ and Panjabi MM. (1991). The intersegmental and multisegmental muscles of the lumbar spine: a biomechanical model comparing lateral stabilizing potential. Spine. 16(7): 793-799.
- Jose AM. (2001). Anatomy and Leonardo da Vinci. The Yale journal of biology and medicine. 74(3): 185-195.
- Sanan A, Rengachary SS. (1996).The history of spinal biomechanics. Neurosurgery. 39(4): 657-669.
- Nakatsukasa M, Hayama S and Preuschoft H. (1995). Postcranialskeleton of a macaque trained for bipedal standing and walking and implications for functional adaptation. Folia Primatologica. 64(1-2):1-29.
- Rosenberg K and Trevathan W. (1995). Bipedalism and human birth: The obstetrical dilemma revisited. Evolutionary Anthropology: Issues, News, and Reviews. 4(5):161- 168.
- Whitcome KK, Shapiro LJ and Lieberman DE. (2007). Fetal load and the evolution of lumbar lordosis in bipedal hominins. Nature. 450(7172): 1075-1078.
- Bruner E, Grimaud-Hervé D, Wu X, Holloway R, et al. (2015). A paleoneurological survey of Homo erectus endocranial metrics. Quaternary International. 368: 80-87.
- Zhang Y, Wu X and Schepartz LA. (2015). Comparing methods for estimating cranial capacity in incomplete Human fossils using the Jingchuan 1 partial cranium as an example Quaternary International. 434: 57-64.
- Stringer C. (2016). The origin and evolution of Homo sapiens. Phil. Trans. R. Soc.B. 371 (1698): 20150237.
- Matsuzawa T. (2009). Symbolic representation of number in chimpanzees. Current opinion in neurobiology. 19(1): 92-98.